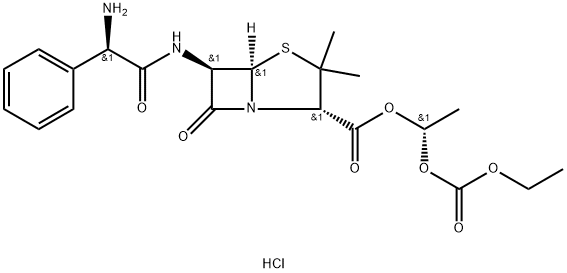
BACAMPICILLIN
- CAS NO.:50972-17-3
- Empirical Formula: C21H27N3O7S
- Molecular Weight: 465.52
- MDL number: MFCD00864977
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:58:12
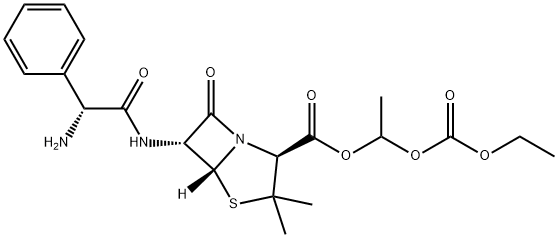
What is BACAMPICILLIN?
Absorption
Absorbed following oral administration.
Originator
Penglobe,Astra,W. Germany,1977
Background
Bacampicillin is a prodrug of ampicillin and is microbiologically inactive. It is absorbed following oral administration. During absorption from the gastrointestinal tract, bacampicillin is hydrolyzed by esterases present in the intestinal wall. It is microbiologically active as ampicillin, and exerts a bactericidal action through the inhibition of the biosynthesis of cell wall mucopeptides. It is used to cure infection of upper and lower respiratory tract; skin and soft tissue; urinary tract and acute uncomplicated gonococcal urethritis etc.
Indications
For infections at the following sites: upper and lower respiratory tract; skin and soft tissue; urinary tract and acute uncomplicated gonococcal urethritis, when due to sensitive strains of the following organisms: Gram-positive: streptococci (including S. faecalis and S. pneumoniae) and nonpenicillinase-producing staphylococci; Gram-negative: H. influenzae, N. gonorrhoeae, E. coli, P. mirabilis, Salmonellae and Shigellae.
Definition
ChEBI: A penicillanic acid ester that is the 1-ethoxycarbonyloxyethyl ester of ampicillin. It is a semi-synthetic, microbiologically inactive prodrug of ampicillin.
Manufacturing Process
1'-Ethoxycarbonyloxyethyl 6-(D-α-azidophenylacetamido)penicillinate (98 g)
was prepared from sodium 6-(D-α-azidophenylacetamido)penicillinate (397 g,
1 mol), α-chlorodiethylcarbonate (458 g, 3 mols) and sodium bicarbonate
(504 g, 6 mols). The product showed strong IR absorption at 2090 cm-1 and
1780-1750 cm-1 showing the presence of azido group and β-lactam and ester
carbonyls.
It was dissolved in ethyl acetate (700 ml) and hydrogenated at ambient
conditions over a palladium (5%)on carbon catalyst (18 g). The catalyst was
removed by filtration and washed with ethyl acetate. The combined filtrates
were extracted with water at pH 2.5 by addition of dilute hydrochloric acid.
Lyophilization of the aqueous phase gave the hydrochloride of 1'-
ethoxycarbonyloxyethyl 6-(D-α-aminophenylacetarnido)penicillinate (94 g), MP
171°-176°C.
Therapeutic Function
Antibacterial
Pharmacokinetics
Bacampicillin is a prodrug of ampicillin and is microbiologically inactive.
Metabolism
Not Available
Metabolism
Although comparatively well absorbed (30–55%), the oral efficacy of ampicillin for systemic infections can be enhanced significantly through the preparation of pro-drugs. In contrast to ampicillin itself, which is amphoteric, bacampicillin is a weak base and is very well absorbed in the duodenum (80–98%). Enzymatic ester hydrolysis in the gut wall liberates carbon dioxide and ethanol, followed by spontaneous loss of acetaldehyde and production of ampicillin. The acetaldehyde is metabolized oxidatively by alcohol dehydrogenase to produce acetic acid, which joins the normal metabolic pool.
Properties of BACAMPICILLIN
| Boiling point: | 678.4±55.0 °C(Predicted) |
| Density | 1.37±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | Soluble in DMSO |
| pka | pKa 6.8 (Uncertain) |
| CAS DataBase Reference | 50972-17-3 |
Safety information for BACAMPICILLIN
Computed Descriptors for BACAMPICILLIN
New Products
Tridecanoic acid 3-Bromophenylacetic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 3′-Hydroxyacetophenone 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline Methyl-5-chloro-2-hdyroxy-3-nitro benzoate 5-(2,4-Difluorophenyl) isoxazole-3-carboxylicacid Methyl-5-(3-nitrophenyl)isoxazole-3-carboxylate 2-Bromo-1-(5-fluoro-2-hydroxyphenyl)ethanone Methl-4-(chlorophenyl)-2,4-dioxo butanoate 2-Morpholino nicotinicacid 5-amino-1-(2-chlorophenyl)-1H-pyrazole-4-carbonitrile ethyl 1-(6-chloropyridin-2-yl)-5-hydroxy-1H-pyrazole-4-carboxylate N-[-6,6-dimethyl-2-hepten-4-yn-1-yl]-N-(methyl-d3)-1-naphthalenemethanamine, monohydrochloride 2H-indol-2-one, 5-amino-1,3-dihydro-1-(1-methylethyl)- Nifedipine D6Related products of tetrahydrofuran


You may like
-
 (3-(4-Chlorophenyl)-1-phenyl-1H-pyrazol-4-yl)methanol 98%View Details
(3-(4-Chlorophenyl)-1-phenyl-1H-pyrazol-4-yl)methanol 98%View Details
36640-39-8 -
 32332-98-2 98%View Details
32332-98-2 98%View Details
32332-98-2 -
 4214-73-7 2-Amino-5-cyanopyridine 98+View Details
4214-73-7 2-Amino-5-cyanopyridine 98+View Details
4214-73-7 -
 139585-70-9 98+View Details
139585-70-9 98+View Details
139585-70-9 -
 4214-73-7 2-Amino-5-cyanopyridine 98+View Details
4214-73-7 2-Amino-5-cyanopyridine 98+View Details
4214-73-7 -
 139585-70-9 98+View Details
139585-70-9 98+View Details
139585-70-9 -
 4214-73-7 2-Amino-5-cyanopyridine 98+View Details
4214-73-7 2-Amino-5-cyanopyridine 98+View Details
4214-73-7 -
 139585-70-9 98+View Details
139585-70-9 98+View Details
139585-70-9
