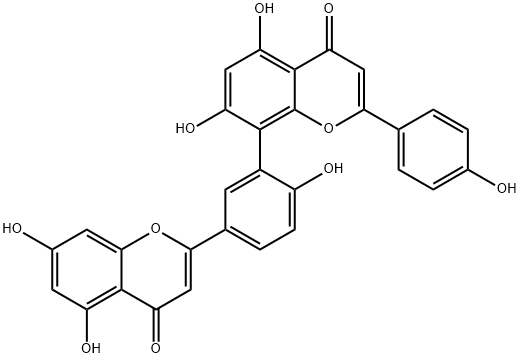
Amentoflavone
Synonym(s):Didemethyl-ginkgetin;I3′,II8-Biapigenin;Tridemethylsciadopitysin
- CAS NO.:1617-53-4
- Empirical Formula: C30H18O10
- Molecular Weight: 538.46
- MDL number: MFCD00017470
- EINECS: 000-000-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-13 23:13:44
What is Amentoflavone?
Description
Amentoflavone is a biflavonoid originally isolated from Selaginella. It has a wide variety of biological effects including antibacterial, antioxidant, antiviral, antidiabetic, and neuroprotective activities. Amentoflavone has antiviral activity against the influenza A subtypes H1N1 and H3N2, influenza B, and herpes simplex virus 1 (EC50s = 3.1 and 4.3, 0.56, and 17.9 μg/ml, respectively). It has antidiabetic effects such that it dose-dependently increases insulin receptor phosphorylation and activation and inhibits hydrolysis of p-nitrophenyl phosphate (p-NPP) catalyzed by protein tyrosine phosphatase 1B (PTP1B; IC50 = 7.3 μM). Amentoflavone reduces the time mice spend immobile in the forced swim test, a measure of antidepressant efficacy, in a dose-dependent manner.
Description
Amentoflavone is a mild antidepressant found in many plants, such as?Hypericum perforatum?L. (St. John''s wort). It has also been shown to exhibit antiviral and anticancer activity and is a potent antioxidant.
The Uses of Amentoflavone
Amentoflavone, is isolated from an Et acetate ext. of the whole plant of Selaginella tamariscina. It has been shown to have antitumor activity, such as mitochondria-mediated apoptotic cell death. Amentoflavone can interact with many medications by being a potent inhibitor of CYP3A4 and CYP2C9, which are enzymes responsible for the metabolism of some drugs in the body.
What are the applications of Application
Amentoflavone is an anti-viral and anti-inflammatory agent that up-regulates PPAR-γ
Definition
ChEBI: Amentoflavone is a biflavonoid that is obtained by oxidative coupling of two molecules of apigenin resulting in a bond between positions C-3 of the hydroxyphenyl ring and C-8 of the chromene ring. A natural product found particularly in Ginkgo biloba and Hypericum perforatum. It has a role as a cathepsin B inhibitor, an antiviral agent, an angiogenesis inhibitor, a P450 inhibitor and a plant metabolite. It is a biflavonoid, a hydroxyflavone and a ring assembly.
Biochem/physiol Actions
Biflavonoid with anti-inflammatory, anti-viral and cancer chemopreventive activity. It inhibits vascularization of tumors by blocking the activity of angiogenic VEGFs. Blocks the induction of COX-2 and up-regulates PPAR-γ. It is a negative modulator of the GABAA receptor at the benzodiazepine binding site.
Properties of Amentoflavone
| Melting point: | >300°C (dec.) |
| Boiling point: | 910.5±65.0 °C(Predicted) |
| Density | 1.656±0.06 g/cm3(Predicted) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | DMSO (Slightly), Methanol (Slightly, Heated), Pyridine (Slightly) |
| form | neat |
| pka | 6.01±0.40(Predicted) |
| form | Solid |
| color | Yellow |
| BRN | 380244 |
| CAS DataBase Reference | 1617-53-4(CAS DataBase Reference) |
Safety information for Amentoflavone
Computed Descriptors for Amentoflavone
| InChIKey | YUSWMAULDXZHPY-UHFFFAOYSA-N |
New Products
Methyl N-((2'-cyano-[1,1'-biphenyl]-4-yl)methyl)-N-nitroso-L-valinate Levofloxacin - Impurity B/ Levofloxacin Related Compound A Ivabradine R isomer Escitalopram - Impurity K Escitalopram Cyno dilol Impurity Paroxetine EP impurity I/Paroxetine Anhydrous EP Impurity I 2-Amino-5-Bromo-6-Chloropyridine 3-Amino-5-bromopicolinic acid 1-(7,8-Dinitro-4,5-dihydro-1H-1,5-methanobenzo[d]azepin-3(2H)-yl)-2,2,2-trifluoroethanone 2-Amino-3-Bromo-5-Cyanopyridine 2-Amino-3-Chloropyridine 2,5-Dibromo-3-MethylpyridineRelated products of tetrahydrofuran


You may like
-
 Amentoflavone CAS 1617-53-4View Details
Amentoflavone CAS 1617-53-4View Details
1617-53-4 -
 Amentoflavone CAS 1617-53-4View Details
Amentoflavone CAS 1617-53-4View Details
1617-53-4 -
 Amentoflavone CAS 1617-53-4View Details
Amentoflavone CAS 1617-53-4View Details
1617-53-4 -
 17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 -
 Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
120279-90-5 -
 76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 -
 118-23-0 NLT 95%View Details
118-23-0 NLT 95%View Details
118-23-0 -
 573704-41-3 NLT 95%View Details
573704-41-3 NLT 95%View Details
573704-41-3
