4-AMINO-1,8-NAPHTHALIMIDE
- CAS NO.:1742-95-6
- Empirical Formula: C12H8N2O2
- Molecular Weight: 212.2
- MDL number: MFCD00006921
- EINECS: 217-110-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-12 09:17:10
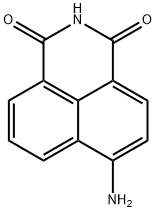
What is 4-AMINO-1,8-NAPHTHALIMIDE?
Description
The poly(ADP-ribose) polymerases (PARPs) form a family of enzymes with roles in DNA repair and apoptosis, particularly in response to reactive oxygen and nitrogen species. 4-amino-1,8-Naphthalimide (4-ANI) is an inhibitor of PARP (IC50 = 180 nM). It blocks radiation-induced PARP in cancer cells, potentiating the cytotoxicity of γ-radiation, although it is not cytotoxic in the absence of radiation. 4-ANI is used to study the role of PARP activity in various cell systems.
Chemical properties
orange amorphous powder
The Uses of 4-AMINO-1,8-NAPHTHALIMIDE
The poly(ADP-ribose) polymerases (PARPs) form a family of enzymes with roles in DNA repair and apoptosis, particularly in response to reactive oxygen and nitrogen species. 4-amino-1,8-Naphthalimide (4-ANI) is an inhibitor of PARP (IC50 = 180 nM). It blocks radiation-induced PARP in cancer cells, potentiating the cytotoxicity of γ-radiation, although it is not cytotoxic in the absence of radiation. 4-ANI is used to study the role of PARP activity in various cell systems.
What are the applications of Application
4-Amino-1,8-naphthalimide is a PARP-1 inhibitor
Definition
ChEBI: 4-amino-1,8-naphthalimide is a benzoisoquinoline and a dicarboximide.
Biochem/physiol Actions
4-Amino-1,8-naphthalimide sensitizes cells to radiation-induced cell damage and enhances the cytotoxicity of 1-methyl-3-nitro-1-nitrosoguanidine.
Enzyme inhibitor
This PARP inhibitor (FW = 212.21 g/mol) is a strong inhibitor of NAD:ADP-ribosyltransferase or poly(ADP-ribose) polymerase (IC50 = 0.18 μM). In addition, 4-Amino-1,8-naphthalimide is a radiation sensitizer at non-toxic and low concentrations.
References
[1]. schlicker a, peschke p, bürkle a, et al. 4-amino-1,8-naphthalimide: a novel inhibitor of poly(adp-ribose) polymerase and radiation sensitizer. int j radiat biol. 1999 jan;75(1):91-100.
[2]. davar d1, beumer jh, hamieh l, et al. role of parp inhibitors in cancer biology and therapy. curr med chem. 2012;19(23):3907-21.
[3]. banasik m, komura h, shimoyama m,et al. specific inhibitors of poly(adp-ribose) synthetase and mono(adp-ribosyl)transferase. j biol chem. 1992 jan 25;267(3):1569-75.
[4]. issaeva n, thomas hd, djureinovic t, et al. 6-thioguanine selectively kills brca2-defective tumors and overcomes parp inhibitor resistance. cancer res. 2010 aug 1;70(15):6268-76.
Properties of 4-AMINO-1,8-NAPHTHALIMIDE
| Melting point: | 360 °C |
| Boiling point: | 352.04°C (rough estimate) |
| Density | 1.105 g/mL at 25 °C(lit.) |
| vapor pressure | 0Pa at 25℃ |
| refractive index | 1.6000 (estimate) |
| storage temp. | -20°C |
| solubility | Soluble in DMSO |
| form | Orange solid. |
| pka | 9.53±0.20(Predicted) |
| color | Yellow to brown |
| Water Solubility | 354mg/L at 25℃ |
| CAS DataBase Reference | 1742-95-6 |
| EPA Substance Registry System | 1H-Benz[de]isoquinoline-1,3(2H)-dione, 6-amino- (1742-95-6) |
Safety information for 4-AMINO-1,8-NAPHTHALIMIDE
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 4-AMINO-1,8-NAPHTHALIMIDE
New Products
Pentadecanoic acid 3-Bromophenylacetic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 3′-Hydroxyacetophenone 5-fluoro-1,3-benzodioxole 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 2-Bromo-6-fluoroaniline 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole N-(4-Bromophenyl)-2-chloroacetamide 6-Isopropylpicolinonitrile N-Methyl-3,5-dinitro-2-pyridinamine Phenyl-boronic acid-d5 10,11-Dihdyro-10-oxo-5H-dibenz[b,f]azepine-5-carboxamide-d4 5-(2,4-difluorophenyl)oxazolidin-2-oneRelated products of tetrahydrofuran


![2-AMINO-6-NITRO-BENZO[DE]ISOQUINOLINE-1,3-DIONE](https://img.chemicalbook.in/StructureFile/ChemBookStructure2/GIF/CB4477554.gif)
![2-(6-NITRO-1,3-DIOXO-1H-BENZO[DE]ISOQUINOLIN-2(3H)-YL)BENZOIC ACID](https://img.chemicalbook.in/StructureFile/ChemBookStructure3/GIF/CB7330919.gif)
![2-BENZYL-6-PYRROLIDIN-1-YL-1H-BENZO[DE]ISOQUINOLINE-1,3(2H)-DIONE](https://img.chemicalbook.in/StructureFile/ChemBookStructure5/GIF/CB1121599.gif)
![2-(4-AMINOPHENYL)-6-NITRO-1H-BENZO[DE]ISOQUINOLINE-1,3(2H)-DIONE](https://img.chemicalbook.in/StructureFile/ChemBookStructure7/GIF/CB4676924.gif)
You may like
-
 4-Amino-1,8-naphthalimide CAS 1742-95-6View Details
4-Amino-1,8-naphthalimide CAS 1742-95-6View Details
1742-95-6 -
 4-Amino-1,8-NaphthalimideView Details
4-Amino-1,8-NaphthalimideView Details
1742-95-6 -
 Deuterated Styrene-d8 97-99%View Details
Deuterated Styrene-d8 97-99%View Details
19361-62-7 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 -
 32659-49-7 97-99%View Details
32659-49-7 97-99%View Details
32659-49-7
