4-Acetoxyindole
Synonym(s):4-Acetoxyindole
- CAS NO.:5585-96-6
- Empirical Formula: C10H9NO2
- Molecular Weight: 175.18
- MDL number: MFCD00010678
- EINECS: 1312995-182-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-28 03:01:26
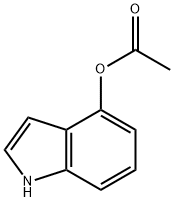
What is 4-Acetoxyindole?
Description
4-acetoxylindole is also known as 4-Indolyl acetate. It can be used as standards of the reverse phase HPLC during separation of small molecules. It can also be used as reactant during the preparation of inhibitors of HIV-attachment, prostanoid EP3 antagonist, indolyl-nitroalkanes and psilocybin.
The Uses of 4-Acetoxyindole
4-Acetoxyindole is a useful intermediate for organic synthesis of Psilocine and Psilocine analogs.
The Uses of 4-Acetoxyindole
Reactant for preparation of:• ;Inhibitors of HIV-1 attachment1• ;Prostanoid EP3 receptor antagonist, as a novel antiplatelet agent2• ;Indolyl-nitroalkanes via N-bromosuccinimide catalyzed synthesis3• ;Psilocybin as a medicinal agent4
The Uses of 4-Acetoxyindole
Reactant for preparation of:
- Inhibitors of HIV-1 attachment
- Prostanoid EP3 receptor antagonist, as a novel antiplatelet agent
- Indolyl-nitroalkanes via N-bromosuccinimide catalyzed synthesis
- Psilocybin as a medicinal agent
General Description
4-Acetoxyindole is a chromophore that belongs to the pyrrole family of compounds. It has been shown to react with an ionic liquid under acidic conditions to form a protonated intermediate, which can be deprotonated by a nucleophile. This reaction yields an acetate anion and a fluorescing product. 4-Acetoxyindole also reacts with deuterium gas, yielding an acetate, a deuterium atom, and fluorescing product. The reaction is reversible and the yield of the product depends on the concentration of the reactants. 4-Acetoxyindole has strong carbonyl groups that make it reactive towards other functional groups. These reactions are useful for synthesizing heterocycles such as indoles and isoquinolines.
References
http://www.sigmaaldrich.com/catalog/product/aldrich/259047?lang=en®ion=US
https://pubchem.ncbi.nlm.nih.gov/compound/4-Acetoxyindole
Properties of 4-Acetoxyindole
| Melting point: | 100-102°C |
| Boiling point: | 339.1±15.0 °C(Predicted) |
| Density | 1?+-.0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) |
| form | Solid |
| pka | 16.38±0.30(Predicted) |
| color | Off-White to Beige |
| InChI | InChI=1S/C10H9NO2/c1-7(12)13-10-4-2-3-9-8(10)5-6-11-9/h2-6,11H,1H3 |
| CAS DataBase Reference | 5585-96-6(CAS DataBase Reference) |
Safety information for 4-Acetoxyindole
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H332:Acute toxicity,inhalation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P310:Immediately call a POISON CENTER or doctor/physician. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 4-Acetoxyindole
| InChIKey | ZXDMUHFTJWEDEF-UHFFFAOYSA-N |
| SMILES | N1C2=C(C(OC(=O)C)=CC=C2)C=C1 |
New Products
Pentadecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetrahydropyranyl-4-acetic acid Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran


You may like
-
 5585-96-6 4-Indoxyl acetate 98%View Details
5585-96-6 4-Indoxyl acetate 98%View Details
5585-96-6 -
 5585-96-6 4-acetoxyindole 98%View Details
5585-96-6 4-acetoxyindole 98%View Details
5585-96-6 -
 4-Indolyl acetate CAS 5585-96-6View Details
4-Indolyl acetate CAS 5585-96-6View Details
5585-96-6 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
