1,6-NAPHTHYRIDINE
- CAS NO.:253-72-5
- Empirical Formula: C8H6N2
- Molecular Weight: 130.15
- MDL number: MFCD00059750
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:59:02
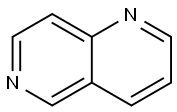
What is 1,6-NAPHTHYRIDINE?
Description
Naphthalene is an aromatic organic compound that consists of two fused benzene rings. Diazanaphthalenes have the naphthalene structure with nitrogen atoms in place of two of the CH groups. There are two structural classes of diazanaphthalenes: benzodiazines, in which both nitrogen atoms are in the same ring, and naphthyridines, with one nitrogen atom in each ring.
Six naphthyridine isomers exist, based on the positions of the nitrogen atoms; they can be in the 1,5, 1,6 (shown here), 1,7, 1,8, 2,6, or 2,7 positions. All are white solids with a surprisingly wide span of melting points: 1,6-Naphthyridine’s is the lowest at <40 oC; 2,6-naphthyridine’s is the highest at 114–115 oC.
Quinoline derivatives such as naphthyridines that have nitrogen atoms in the 1-position are usually synthesized via the Skraup reaction, in which an aminopyridine and a glycerol derivative are heated in the presence of an oxidizing agent (e.g., nitrobenzene) and sulfuric acid. This is often a violent, runaway reaction, so it is perhaps fortunate that the synthesis of 1,6-naphthyridine from 4-aminopyridine was not originally successful.
Refinements of the Skraup reaction, however, did eventually lead to preparations of 1,6-naphthyridine in modest yields. One modification used 4-aminopyridine-N-oxide as the starting material; the resulting 1,6-naphthyridine-N-oxide was then reduced to the free base.
1,6-Naphthyridine and some of its derivatives have been reported to have medicinal, electronic, and catalytic properties. But none of these investigations has yet resulted in any practical applications.
Definition
ChEBI: 1,6-naphthyridine is a naphthyridine.
Properties of 1,6-NAPHTHYRIDINE
| Melting point: | 231-233°C |
| Boiling point: | 267.3±13.0 °C(Predicted) |
| Density | 1.183±0.06 g/cm3(Predicted) |
| Flash point: | 35 °C |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | unreported |
| form | powder to crystal |
| appearance | white to pale yellow or light brown solid or liquid |
| pka | 3.70±0.10(Predicted) |
| color | White to Light yellow |
| CAS DataBase Reference | 253-72-5(CAS DataBase Reference) |
| EPA Substance Registry System | 1,6-Naphthyridine (253-72-5) |
Safety information for 1,6-NAPHTHYRIDINE
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H318:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 1,6-NAPHTHYRIDINE
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran


![3-[(E)-3-(DIMETHYLAMINO)-2-PROPENOYL]-2-METHYL-1,6-NAPHTHYRIDINE](https://img.chemicalbook.in/StructureFile/ChemBookStructure2/GIF/CB8402581.gif)



You may like
-
 1,6-Naphthyridine CAS 253-72-5View Details
1,6-Naphthyridine CAS 253-72-5View Details
253-72-5 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
