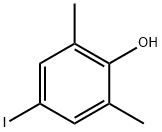
1-Iodo-3,5-dimethylbenzene
Synonym(s):5-Iodo-m-xylene
- CAS NO.:22445-41-6
- Empirical Formula: C8H9I
- Molecular Weight: 232.06
- MDL number: MFCD00060659
- EINECS: 629-121-2
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-01-13 11:13:54
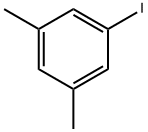
What is 1-Iodo-3,5-dimethylbenzene?
Chemical properties
clear light yellow liquid
The Uses of 1-Iodo-3,5-dimethylbenzene
1-Iodo-3,5-dimethylbenzene (5-iodo-m-xylene) is suitable for use in the synthesis of N-(3,5-xylyl)-N-ethylaniline, an arylamine.
It may be used in the following studies:
- α-Arylation of ketones.
- Copper-catalyzed N-arylation of imidazoles.
- Cyanation of 5-iodo-m-xylene to form 3,5-dimethylbenzonitrile.
- Synthesis of 1,3-Dimethyl-5-phenoxybenzene by nano-CuFe2O4 catalyzed C-O cross-coupling with phenol.
- CuBr-catalyzed amination of 1-iodo-3,5-dimethylbenzene to form N-Allyl-3,5-dimethylbenzenamine.
- Copper-catalyzed C-S bond-formation between 5-iodo-m-xylene and thiophenol.
- As a starting material in the synthesis of biphenyl-3,3′,5,5′-tetracarboxylic acid.
- Radical bromination of 5-iodo-m-xylene by N-bromosuccinimide to form 1,3-bis(bromomethyl)-5-iodobenzene.
The Uses of 1-Iodo-3,5-dimethylbenzene
suzuki reaction
The Uses of 1-Iodo-3,5-dimethylbenzene
1-Iodo-3,5-dimethylbenzene (5-iodo-m-xylene) is suitable for use in the synthesis of N-(3,5-xylyl)-N-ethyl aniline, an aryl amine, It is also used in the following studies; α-Arylation of ketones, Copper-catalyzed N-arylation of imidazoles, Cyanation of 5-iodo-m-xylene to form 3,5-dimethylbenzonitrile, synthesis of 1,3-Dimethyl-5-phenoxybenzene by nano-CuFe2O4 catalyzed C-O cross-coupling with phenol, CuBr-catalyzed amination of 1-iodo-3,5-dimethylbenzene to form N-Allyl-3,5-dimethylbenzenamine, copper-catalyzed C-S bond-formation between 5-iodo-m-xylene and thiophenol. It is also employed as a starting material in the synthesis of biphenyl-3,3?,5,5?-tetracarboxylic acid, radical bromination of 5-iodo-m-xylene by N-bromosuccinimide to form 1,3-bis(bromomethyl)-5-iodobenzene.
General Description
1-Iodo-3,5-dimethylbenzene (5-Iodo-m-xylene) is an aryl halide. It can be obtained from 5-bromo-m-xylene, via copper-catalyzed halogen exchange reaction, in the presence of NaI or KI in n-BuOH or DMF (solvents). It undergoes reaction with phenol in the presence of CuFe2O4 nano powder as a recyclable catalyst to afford 1,3-dimethyl-5-phenoxybenzene.
Properties of 1-Iodo-3,5-dimethylbenzene
| Melting point: | -2.9°C (estimate) |
| Boiling point: | 92-94 °C3 mm Hg(lit.) |
| Density | 1.608 g/mL at 25 °C(lit.) |
| refractive index | n |
| Flash point: | >230 °F |
| storage temp. | Keep in dark place,Sealed in dry,Room Temperature |
| solubility | Sparingly soluble (0.014 g/L) (25°C), Calc. |
| form | Liquid |
| color | Clear light yellow |
| Specific Gravity | 1.608 |
| Sensitive | Light Sensitive |
| BRN | 2039353 |
| CAS DataBase Reference | 22445-41-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Benzene, 1-iodo-3,5-dimethyl-(22445-41-6) |
Safety information for 1-Iodo-3,5-dimethylbenzene
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 1-Iodo-3,5-dimethylbenzene
| InChIKey | ZLMKEENUYIUKKC-UHFFFAOYSA-N |
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate 3,6-Dichloropyridazine Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane 4-Morpholinoaniline 2-Chloromethyl-6-methyl-pyridine 1-Indanone tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 2-Pyridinecarboxaldehdye 6-Methyl-2-pyridinemethanol 4-Bromo-2-fluoro-N-methylbenzamide 3-Azetidinecarboxylic acid N-(2-(4-((4-Fluorobenzyl)carbamoyl)-5-hydroxy-6-methoxypyrimidin-2-yl)propan-2-yl)-5-methyl-1,3,4-oxadiazole-2-carboxamide (R)-4-Boc-morpholine-3-carboxylic acid (Required high chiral purity NLT:99.5%) Tetrahydro-3-(2-methyl-2- nitropropyl)-2H-pyran-2-one 3-Bromo-2-fluorobenzonitrile Desoximetasone Boron Triiodide Nicotinic acid RAPAMYCIN Pentachlorobenzonitrile BudesonideRelated products of tetrahydrofuran



You may like
-
 5-Iodo-m-xylene CAS 22445-41-6View Details
5-Iodo-m-xylene CAS 22445-41-6View Details
22445-41-6 -
 1-Iodo-3,5-dimethylbenzene CAS 22445-41-6View Details
1-Iodo-3,5-dimethylbenzene CAS 22445-41-6View Details
22445-41-6 -
 2-Chlorophenyl diphenyl chloromethane 99%View Details
2-Chlorophenyl diphenyl chloromethane 99%View Details
42074-68-0 -
 6-(chloromethyl)pteridine-2,4-diamine monohydrochloride 82778?08?3 99%View Details
6-(chloromethyl)pteridine-2,4-diamine monohydrochloride 82778?08?3 99%View Details
82778?08?3 -
 1,3-Dibromo-5-fluorobenzene 99%View Details
1,3-Dibromo-5-fluorobenzene 99%View Details
1435-51-2 -
![729559-09-5 [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 729559-09-5 [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 99%View Details
729559-09-5 [N(E)]-N-Ethylidene-2-methyl-2-propanesulfinamide 99%View Details
729559-09-5 -
 4-Isopropyl Thiophenol 4946-14-9 99%View Details
4-Isopropyl Thiophenol 4946-14-9 99%View Details
4946-14-9 -
 2709271-25-8 99%View Details
2709271-25-8 99%View Details
2709271-25-8
