Supplier Type
- Manufacturer
- Reagent
- Trader
- custom synthesis
- contract manufacturer
Supplier Region
- Hyderabad(35)
- Gujarat(54)
- Maharashtra(37)
- Bangalore(3)
- Tamil Nadu(7)
- Mumbai(71)
- New Delhi(6)
- Vadodara(16)
- Rajasthan(1)
- Andhra Pradesh(5)
- Beijing(5)
- Delhi(12)
- Ahmedabad(17)
- Uttar Pradesh(7)
- Telangana(3)
- Pune(6)
- Nagpur(2)
- Karnataka(6)
- Kolkata(4)
- Madhya Pradesh(4)
- West Bengal(1)
- Himachal Pradesh(1)
- Haryana(2)
Purity
- Zn 27% min.
- We offer Acephate with 97% min purity which is available in SP(Water Soluble Powder) and WP(Wettable
- Salicylic Acid
- Rasagiline Mesylate Impurity C
- Pure
- NLT 99.5%
- NLT 95%
- NLT 90%
- more than 95%
- more than 90%
- Mesalazine EP Impurity H
- Mathadone (M225865) impurity. Used in synthesis of methadone, antispasmodics and other pharmaceutica
- Greater than 99%
- Dipropargyl r- amino indane
- Dapagliflozin Intermediate, Dapagliflozin Impurity
- API Standards
- API Impurity / In-house working standard
- Acetylsalicylic Acid EP Impurity C
- ABOVE 99%
- 99.99%
- 99.98%
- 99.97%
- 99.95%
- 99.91%
- 99.90%
- 99.9%
- 99.86%
- 99.85%
- 99.80%
- 99.73%
- 99.70%
- 99.65%
- 99.50%
- 99.5%
- 99.5
- 99.42%
- 99.40%
- 99.29%
- 99% TO 101%
- >99%
- 99%
- 99 %
- 98+
- 98.60%
- 98.5%
- 98.12%
- 98.00%
- 98% MIN
- 98% +
- >98% / >99%
- > 98%
- 98%
- >98%
- 97-99%
- 97.81%
- 97.57%
- 97.51%
- 97.00%
- 97%
- >97%
- 97 %
- 96.80%
- 96.39%
- 96.35%
- 96%
- 95.00%
- >95%
- 95%
- 94.00%
- 93.4%
- >92%
- >90%
- 90%
- 90 % Above
- 80%
- 60%
- 33.2 %
- 33%
- 30%
- 28%
- 25%
- 25 MG 50 MG 100 MG 250 MG EXTRA
- 21%
- 12%/15%
- 100%
- (R)-N,N-di(prop-2-yn-1-yl)-2,3- dihydro-1
Package
- 2.5501e+00925 MG 50 MG 100 MG 250 MG EXTRA
- 1g
- 5g
- 10g
- 25g
- 100ml
- 100g
- 500ml
- 500g
- 1L
- 5000ml
- 50kg
- 100kg
- 500kg
- 95NLT %
- 11Between 1 gm to 1 MT (As per availability)
- 0Jumbo Bags or as per customer requirements
- 25mg
- 50mg
- 100mg
- 250mg
- 500mg
- 1gm
- 50g
- 1kg
- 5kg
- 10kg
- 25kg
- 1MT
- 1and gm
- 50kgs
- 250250 mg.
-
Meta-GliMepiride IMpurity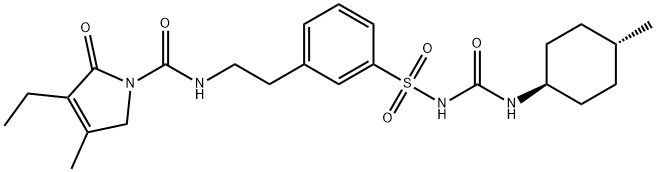
791104-62-6
-
cis-Glimepiride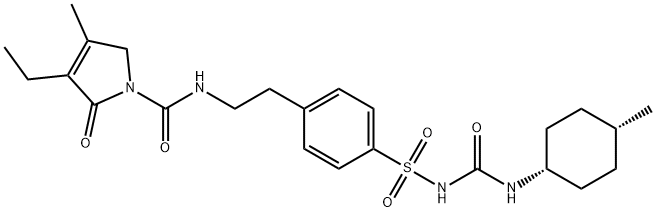
684286-46-2
-
ortho-GliMepiride IMpurity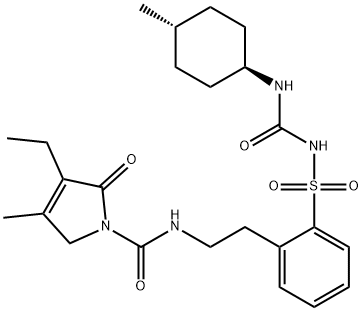
878480-70-7
-
1H-Pyrrole-1-carboxamide, 3-ethyl-2,5-dihydro-4-methyl-N-[2-[4-[[[[(4-methylphenyl)amino]carbonyl]amino]sulfonyl]phenyl]ethyl]-2-oxo-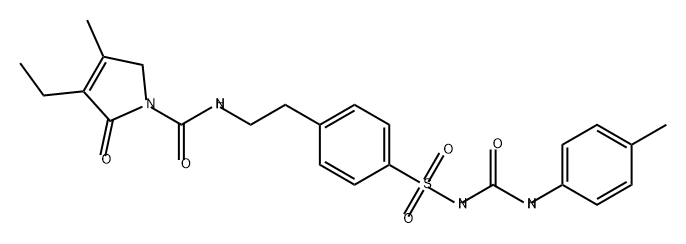
2724503-28-8
-
3,4-dihydro-3-(4-piperidinyl)-2(1H)-Quinolinone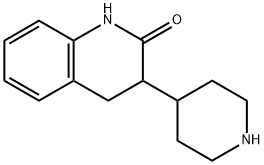
741235-48-3
-
GLIMEPIRIDE INTERMEDIATE STEP-1 (TRIMER IMPURITY)
-
GLIMEPIRIDE INTERMEDIATE STEP-1 (DIMER IMPURITY)
-
4-[2-[(3-Ethyl-4-methyl-2-oxo-3-pyrrolin-1-yl)carboxamido]ethyl]benzenesulfonamide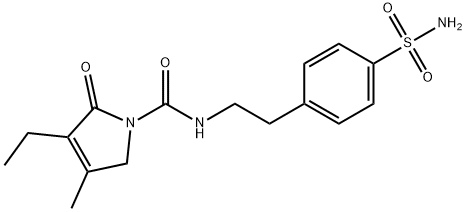
119018-29-0
-
Everolimus Related Compound 2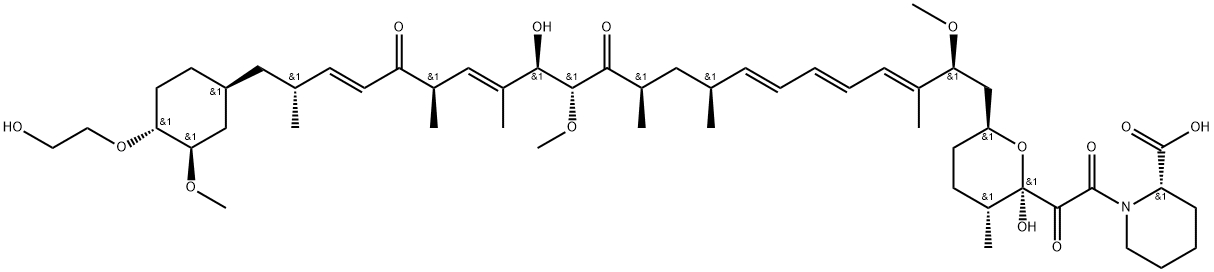
1062122-63-7
-
Lacosamide impurity 8/Lacosamide EP Impurity H/(S)-2-acetamido-N-((S)-1-(benzylamino)-3-methoxy-1-oxopropan-2-yl)-3-methoxypropanamide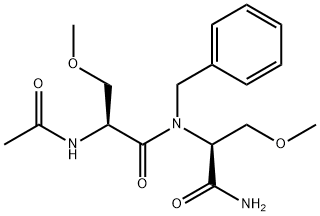
1375090-19-9

N,N-Dimethylacetamide 127-19-5

Aminotri-(methylenephosphonic acid) 6419-19-8
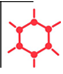
Tetrabutylammonium bromide 1643-19-2

Sodium p-aminosalicylate dihydrate 6018-19-5

Acetic acid 64-19-7

Sodium carbonate 497-19-8

Acetic acid 64-19-7
Irinotecan Impurity L (HCl Salt) 1992961-26-8

Direct black 19 6428-31-5

Sodium carbonate 497-19-8


