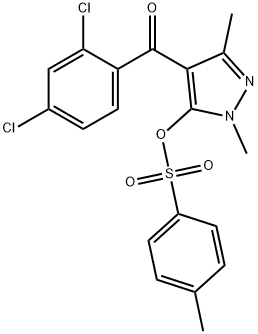
COMPUTED DESCRIPTORS
| Molecular Weight | 439.3 g/mol |
|---|---|
| XLogP3 | 3.8 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Exact Mass | 438.0207836 g/mol |
| Monoisotopic Mass | 438.0207836 g/mol |
| Topological Polar Surface Area | 86.6 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 663 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Pyrazolynate is a member of the class of pyrazoles that is 1,3-dimethylpyrazole which is substituted at positions 4 and 5 by 2,4-dichlorobenzoyl and p-tosyloxy groups, respectively. It is an obsolete proherbicide (via hydrolysis of the tosylate group to afford the corresponding 5-hydroxypyrazole), that was used to control weeds in rice paddy fields. It has a role as a proherbicide, an EC 1.13.11.27 (4-hydroxyphenylpyruvate dioxygenase) inhibitor, an agrochemical and a carotenoid biosynthesis inhibitor. It is a member of pyrazoles, a tosylate ester, an aromatic ketone and a dichlorobenzene.