CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Solid |
|---|---|
| Melting Point | Needles from water; mp: 268 °C (effervescence); specific optical rotation: -258 deg @ 20 °C (c=0.38) for the anhydrous salt /Chloride, l-Form/ |
| Solubility | Solublity (25 °C): approx 50 mg/l water; but supersaturated solustions are formed readily. Solubility also reported as approximately 1 g/40 ml water; approximately 1 g/75 ml ethanol. Also soluble in methanol. Insoluble in pyridine, chloroform, benzene, acetone, ether. /Chloride, d-Form/ |
| Stability/Shelf Life | SENSITIVE TO LIGHT /CHLORIDE/ |
| Optical Rotation | Hexagonal and pentagonal microplatelets from water. Can exist in the form of various hydrates. The anhydrous materl (dec 274-275 °C) takes up water in moist atm until it reaches the pentahydrate stage, dec approximately 270 °C. Specific optical rotation: +190 deg @ 22 °C/D; +219 deg @ 23 °C/D (c= 0.785 in methanol). UV max (H2O): 280 nm (E=118, 1%, 1 cm). /Chloride, d-Form/ |
| Decomposition | When heated to decomposition it emits toxic fumes of nitroxides. |
| Collision Cross Section | 236 Ų [M+H]+ [CCS Type: TW, Method: calibrated with polyalanine and drug standards] |
| Other Experimental Properties | White to light-tan crystalline alkaloid; odorless. MP 270 °C with decomposition. Soluble in water and alcohol; insoluble in acetone, chloroform, and ether; aqueous solution is strongly dextrorotary (specific rotation for 1% solution of anhydrous -208 to +218 degrees). /Chloride/ |
COMPUTED DESCRIPTORS
| Molecular Weight | 609.7 g/mol |
|---|---|
| XLogP3 | 6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 2 |
| Exact Mass | 609.29646203 g/mol |
| Monoisotopic Mass | 609.29646203 g/mol |
| Topological Polar Surface Area | 80.6 Ų |
| Heavy Atom Count | 45 |
| Formal Charge | 1 |
| Complexity | 990 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
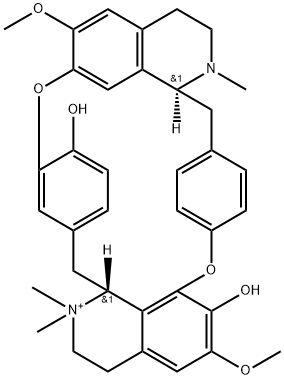
Tubocurarine is a benzylisoquinoline alkaloid muscle relaxant which constitutes the active component of curare. It has a role as a nicotinic antagonist, a muscle relaxant and a drug allergen. It derives from a hydride of a tubocuraran.