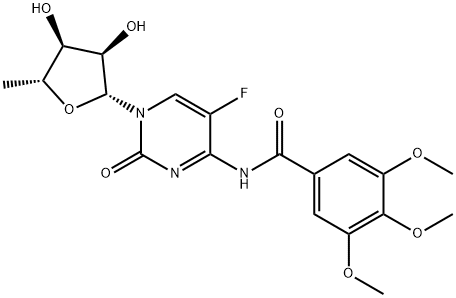
COMPUTED DESCRIPTORS
| Molecular Weight | 439.4 g/mol |
|---|---|
| XLogP3 | -0.1 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 6 |
| Exact Mass | 439.13909283 g/mol |
| Monoisotopic Mass | 439.13909283 g/mol |
| Topological Polar Surface Area | 139 Ų |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 748 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Galocitabine is an orally available 5-fluorouracil (5-FU) prodrug with potential antineoplastic activity. Upon administration, galocitabine is converted into 5'-deoxy-5-fluorocytidine, 5'-deoxy-fluorouridine, and 5-FU. 5-FU is further metabolized into other cytotoxic metabolites that interfere with RNA and DNA synthesis via inhibition of thymidylate synthase. As a result, this agent eventually inhibits tumor cell growth.