Leflunomide
Synonym(s):5-Methyl-N-[4-(trifluoromethyl) phenyl]-isoxazole-4-carboxamide;5-Methylisoxazole-4-(4-trifluoromethyl)carboxanilide;Leflunomide
- CAS NO.:75706-12-6
- Empirical Formula: C12H9F3N2O2
- Molecular Weight: 270.21
- MDL number: MFCD00867593
- EINECS: 616-254-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-04-17 18:50:11
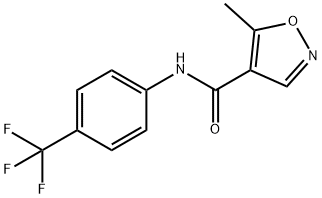
What is Leflunomide?
Absorption
Well absorbed, peak plasma concentrations appear 6-12 hours after dosing
Toxicity
LD50=100-250 mg/kg (acute oral toxicity)
Description
Leflunomide is an orally-available disease-modifying antirheumatic drug
and was launched as Arava in the US for the treatment of rheumatoid arthritis
(RA) ; it is the first and only drug to be indicated to slow down structural joint
damage of RA, so addressing an unmet medical need.
Leflunomide is prepared in 3 steps from the appropriate acetoacetic anilide
using a nitrile oxide- enamine cycloaddition reaction to assemble the isoxazole
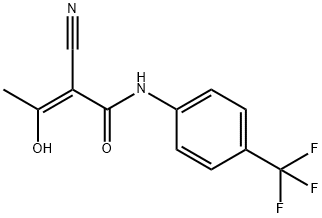
ring. Leflunomide is a prodrug, being extensively metabolized in vivo into the
corresponding 2-cyano-3-hydroxy-2-butenamide resulting from fragmentation of
the isoxazole ring. This cyanoenol is actually the active metabolite and several
experiments in animals have demonstrated that after oral administration,
substantial and sustained levels of this metabolite were delivered to the
systemic circulation.
In vitro, Leflunomide’s active metabolite inhibits dihydroorotate dehydrogenase,
an enzyme involved in the biosynthesis of pyrimidine nucleotides, probably
accounting for its immunosuppressive effect in vivo. Other mechanisms of action
such as inhibition of tyrosine kinase and inhibition of responsiveness to
interleukin-2 have been proposed. In diverse models of autoimmune or allergic
diseases, Leflunornide showed efficacy both prophylactically and therapeutically.
The Uses of Leflunomide
By virtue of its immunosuppressant effects, leflunomide has found use in organ transplantation and treatment of rheumatoid arthritis and other autoimmune diseases.
Background
Leflunomide is a pyrimidine synthesis inhibitor belonging to the DMARD (disease-modifying antirheumatic drug) class of drugs, which are chemically and pharmacologically very heterogeneous. Leflunomide was approved by FDA and in many other countries (e.g., Canada, Europe) in 1999.
Indications
For the management of the signs and symptoms of active rheumatoid arthritis (RA) to improve physical function and to slow the progression of structural damage associated with the disease. Has also been used for the prevention of acute and chronic rejection in recipients of solid organ trasnplants and is designated by the FDA as an orphan drug for this use.
What are the applications of Application
Leflunomide is an inhibitor of Cox-2 and tyrosine kinase
Pharmacokinetics
Leflunomide is a pyrimidine synthesis inhibitor indicated in adults for the treatment of active rheumatoid arthritis (RA). RA is an auto-immune disease characterized by high T-cell activity. T cells have two pathways to synthesize pyrimidines: the salvage pathways and the de novo synthesis. At rest, T lymphocytes meet their metabolic requirements by the salvage pathway. Activated lymphocytes need to expand their pyrimidine pool 7- to 8-fold, while the purine pool is expanded only 2- to 3-fold. To meet the need for more pyrimidines, activated T cells use the de novo pathway for pyrimidine synthesis. Therefore, activated T cells, which are dependent on de novo pyrimidine synthesis, will be more affected by leflunomide's inhibition of dihydroorotate dehydrogenase than other cell types that use the salvage pathway of pyrimidine synthesis.
Metabolism
Primarily hepatic. Leflunomide is converted to its active form following oral intake.
Properties of Leflunomide
| Melting point: | 166.5 °C |
| Boiling point: | 289.3±40.0 °C(Predicted) |
| Density | 1.392±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | Practically insoluble in water, freely soluble in methanol, sparingly soluble in methylene chloride. |
| form | neat |
| color | White |
Safety information for Leflunomide
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H301:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Leflunomide
| InChIKey | VHOGYURTWQBHIL-UHFFFAOYSA-N |
Abamectin manufacturer
Frolic Pharmachem
KARPSCHEM LABORATORIES PVT. LTD.
Dayaram Pharma Chem
Stellar Chemical Laboratories Pvt., Ltd.
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran




You may like
-
 75706-12-6 LEFLUNOMIDE IH 99%View Details
75706-12-6 LEFLUNOMIDE IH 99%View Details
75706-12-6 -
 75706-12-6 96%View Details
75706-12-6 96%View Details
75706-12-6 -
 Leflunomide 5-methyl-N-(4-(trifluoromethyl)phenyl)isoxazole-4-carboxamide 96%View Details
Leflunomide 5-methyl-N-(4-(trifluoromethyl)phenyl)isoxazole-4-carboxamide 96%View Details
75706-12-6 -
 LEFLUNOMIDE 99%View Details
LEFLUNOMIDE 99%View Details -
 75706-12-6 Leflunomide 98%View Details
75706-12-6 Leflunomide 98%View Details
75706-12-6 -
 75706-12-6 98%View Details
75706-12-6 98%View Details
75706-12-6 -
 75706-12-6 98%View Details
75706-12-6 98%View Details
75706-12-6 -
 Leflunomide 75706-12-6 98%View Details
Leflunomide 75706-12-6 98%View Details
75706-12-6