Lamivudine
Synonym(s):Lamivudine;Epivir;Epivir-HBV;Heptovir;Zeffix
- CAS NO.:134678-17-4
- Empirical Formula: C8H11N3O3S
- Molecular Weight: 229.26
- MDL number: MFCD00869739
- EINECS: 603-844-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-06-07 16:07:22
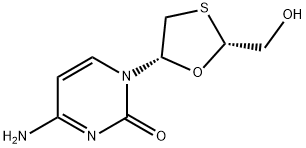
What is Lamivudine?
Absorption
Lamivudine was rapidly absorbed after oral administration in HIV-infected patients. Absolute bioavailability in 12 adult patients was 86% ± 16% (mean ± SD) for the 150-mg tablet and 87% ± 13% for the oral solution. The peak serum lamivudine concentration (Cmax) was 1.5 ± 0.5 mcg/mL when an oral dose of 2 mg/kg twice a day was given to HIV-1 patients. When given with food, absorption is slower, compared to the fasted state.
Toxicity
The most common reported adverse reactions (incidence ≥15%) in adults were headache, nausea, malaise and fatigue, nasal signs and symptoms, diarrhea, and cough.
Description
Lamivudine is a new generation orally active nucleoside analog launched in the U.S.A. for use in combination with zidovudine (AZT) as a first-line therapy for patients with HIV infection. Lamivudine is rapidly converted to phosphorylated metabolites in the body which act as inhibitors and chain terminators of HIV reverse transcriptase (RT), the enzyme required for the replication of the HIV genome. Lamivudine has similar inhibitory potency to RT as AZT but is 10 times less toxic and is active against AZT-resistant strains of HIV.
The Uses of Lamivudine
Lamivudine is used along with other medications to treat human immunodeficiency virus (HIV) infection in adults and children 3 months of age and older.
Lamivudine (Epivir-HBV) is used to treat hepatitis B infection.
Lamivudine is in a class of medications called nucleoside reverse transcriptase inhibitors (NRTIs). It works by decreasing the amount of HIV and hepatitis B in the blood.
Indications
For the treatment of HIV infection and chronic hepatitis B (HBV).
Background
A reverse transcriptase inhibitor and zalcitabine analog in which a sulfur atom replaces the 3' carbon of the pentose ring. It is used to treat Human Immunodeficiency Virus Type 1 (HIV-1) and hepatitis B (HBV).
What are the applications of Application
Lamivudine is a potent reverse transcriptase inhibitor
Pharmacokinetics
Lamivudine is a nucleoside reverse transcriptase inhibitor (NRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1) and hepatitis B (HBV) to disrupt viral DNA synthesis. When phosphorylated, lamivudine can form active metabolites that compete for incorporation into viral DNA. Via DNA incorporation, lamivudine metabolites competitively inhibit the activity of the HIV reverse transcriptase enzyme and act as a chain terminator of DNA synthesis. Due to the lack of a 3'-OH group, incorporated nucleoside analogues prevent the formation of a 5' to 3' phosphodiester linkage that is essential for DNA chain elongation.
Metabolism
Metabolism of lamivudine is a minor route of elimination. In man, the only known metabolite of lamivudine is the trans-sulfoxide metabolite. This biotransformation is catalyzed by sulfotransferases.
Properties of Lamivudine
| Melting point: | 177 °C |
| Boiling point: | 475.4±55.0 °C(Predicted) |
| Density | 1.73±0.1 g/cm3(Predicted) |
| Flash point: | 9℃ |
| storage temp. | 2-8°C |
| solubility | water: soluble10mg/mL, clear |
| form | powder |
| color | white to beige |
| Water Solubility | 70g/L(temperature not stated) |
Safety information for Lamivudine
| Signal word | Warning |
| Pictogram(s) |
 Health Hazard GHS08 |
| GHS Hazard Statements |
H361:Reproductive toxicity |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Lamivudine
| InChIKey | JTEGQNOMFQHVDC-NKWVEPMBSA-N |
Abamectin manufacturer
Jigs Chemical ltd
KRS Pharmaceuticals Pvt. Ltd.
HRV Global Life Sciences
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine N-Benzyl-3-hydroxypiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran



You may like
-
 134678-17-4 98%View Details
134678-17-4 98%View Details
134678-17-4 -
 Lamivudine 98%View Details
Lamivudine 98%View Details -
 134678-17-4 Lamivudine 98%View Details
134678-17-4 Lamivudine 98%View Details
134678-17-4 -
 134678-17-4 98%View Details
134678-17-4 98%View Details
134678-17-4 -
 134678-17-4 Lamivudine 99%View Details
134678-17-4 Lamivudine 99%View Details
134678-17-4 -
 Lamivudine 134678-17-4 98%View Details
Lamivudine 134678-17-4 98%View Details
134678-17-4 -
 134678-17-4 Lamivudine 98%View Details
134678-17-4 Lamivudine 98%View Details
134678-17-4 -
 Lamivudine 134678-17-4 98%View Details
Lamivudine 134678-17-4 98%View Details
134678-17-4