URANYL NITRATE
- CAS NO.:10102-06-4
- Empirical Formula: N2O8U
- Molecular Weight: 394.04
- MDL number: MFCD00036300
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-31 14:38:37
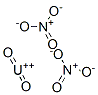
What is URANYL NITRATE?
Chemical properties
Yellow, rhombic crystals. Soluble in water, alcohol, and ether.
Physical properties
The hexahydrate is a yellow crystalline solid; orthogonal crystals; density 2.81 g/cm3; hygroscopic; melts at 60°C; decomposes at 118°C; very soluble in water; soluble in alcohol and ether.
The Uses of URANYL NITRATE
Uranyl nitrate is a yellow poisonous crystal made by dissolving
uranic oxide in nitric acid. It is soluble in water, alcohol, and
ether. First suggested by C. J. Burnett in 1855, a uranyl nitrate
printing process was patented by Niépce de St. Victor in 1858.
The paper was floated on a uranyl nitrate solution and dried
in the dark. The paper was exposed under a negative in the
sun until a faint image appeared. The image was developed to
completion by floating the paper on a solution of silver nitrate
or gold chloride followed by washing. A process patented in
1864 by Jacob Wothly called for adding uranyl nitrate and
silver to collodion, which was then applied to paper. The
Wothlytype was a printing-out process.
Uranyl nitrate was used in printing-out emulsions and with
potassium ferricyanide for toning prints and intensifying negatives.
It was also used with silver nitrate for making uranium/
silver gaslight papers.
The Uses of URANYL NITRATE
Source of uranium dioxide, extraction of uranium into nonaqueous solvents.
Preparation
Uranyl nitrate is obtained as an intermediate in recovering uranium from its minerals. The compound can be prepared by reacting triuranium octaoxide, U3O8, with nitric acid. It is separated and purified by extraction with ether.
Hazard
Strong oxidizer. Corrosive and irritating.
Safety Profile
Poison by inhalation. Moderately toxic by ingestion. Human mutation data reported. A corrosive irritant to skin, eyes, and mucous membranes. A radioactive material. A powerful explosive and oxibzer. Incompatible with cellulose. Ether solutions in sunlight may explode. When heated to decomposition it emits toxic fumes of NOx. See also URANYL NITRATE HEXAHYDRATE and URANIUM.
Properties of URANYL NITRATE
| Melting point: | 60.2°C |
| Boiling point: | 118°C |
| Density | 2.807 |
| solubility | soluble in ethyl ether |
| form | yellow-green solid |
| color | yellow crystals, crystalline |
| Water Solubility | 127 at 25℃ |
| EPA Substance Registry System | Uranyl nitrate (10102-06-4) |
Safety information for URANYL NITRATE
Computed Descriptors for URANYL NITRATE
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5You may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5
