Ubrogepant
- CAS NO.:1374248-77-7
- Empirical Formula: C29H26F3N5O3
- Molecular Weight: 549.54
- MDL number: MFCD28386182
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-14 09:55:04
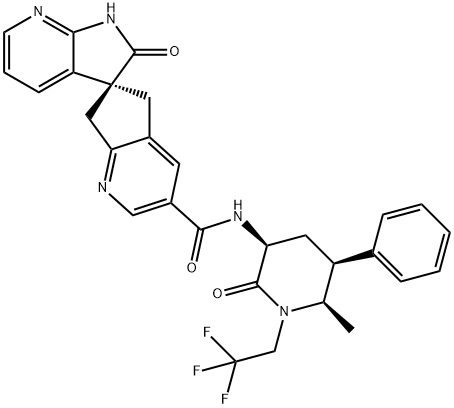
What is Ubrogepant?
Absorption
Following oral administration, Tmax occurs between 0.7 and 1.5 h. When administered with a high-fat meal, Tmax is delayed by approximately 2 hours and Cmax was reduced by 22% with no significant changes to the AUC. Ubrogepant exhibits dose-proportional pharmacokinetics throughout the entirety of its recommended dosing range.
Toxicity
As clinical experience with ubrogepant is limited, detailed toxicity information is not readily available. Prescribing information for ubrogepant recommends a monitoring period of at least 24 hours following overdose based on its 5 to 7 hour half-life.
The Uses of Ubrogepant
Ubrogepant, is a new drug possible used for the treatment of migraine.
Background
Ubrogepant is indicated for the acute treatment of migraine headaches with or without aura in adults. It was approved by the FDA on December 23, 2019, and is the first oral calcitonin gene-related peptide (CGRP) receptor antagonist approved for the acute treatment of migraine. Several oral small molecule CGRP receptor antagonists, belonging to a class of medications referred to as "gepants", have been investigated for migraines, but only ubrogepant and rimegepant remain in clinical development. Previous agents within this class were efficacious but limited by liver toxicity - this led to the development of ubrogepant, which was designed to be a hepatoxicity-free alternative to its predecessors. Several parenteral monoclonal antibodies acting against the CGRP pathway (e.g. erenumab, fremanezumab, galcanezumab) have also been approved in recent years. Ubrogepant was approved by Health Canada on November 10, 2022.
Compared to the current standard of therapy for migraine treatment, namely triptans such as sumatriptan and almotriptan, CGRP antagonists present several advantages. They appear to be better tolerated, do not contribute to medication overuse headaches, and carry no apparent cardiovascular risk, making them suitable for use in patients with cardiovascular disease. The development of oral gepants, including ubrogepant, may therefore constitute a significant advance in migraine headache treatment and may become the new standard of therapy in the treatment of this debilitating condition.
Indications
Ubrogepant is indicated for the acute treatment of migraine with or without aura in adults.
Pharmacokinetics
Ubrogepant acutely treats migraine headache pain by blocking the activity of a key transmitter involved in migraine pathogenesis. Exposure to ubrogepant can be significantly increased in patients with severe hepatic or renal insufficiency - dose adjustments are required for these patients in order to avoid excessive exposure, and ubrogepant is not recommended in patients with end-stage renal disease.
Metabolism
Ubrogepant is eliminated primarily via metabolism, the majority of which is mediated by CYP3A4. Two circulating glucuronide conjugates, along with unchanged parent drug, were found to be the most abundant circulating components in human plasma. The glucuronide metabolites reportedly carry 6000-fold less activity at CGRP receptors and are therefore considered to be pharmacologically inert.
Properties of Ubrogepant
| Boiling point: | 729.4±60.0 °C(Predicted) |
| Density | 1.45±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | Soluble in DMSO |
| form | Solid |
| pka | 11.08±0.20(Predicted) |
| color | White to off-white |
Safety information for Ubrogepant
Computed Descriptors for Ubrogepant
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinYou may like
-
 Ubrogepant 98%View Details
Ubrogepant 98%View Details -
 1374248-77-7 99%View Details
1374248-77-7 99%View Details
1374248-77-7 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
