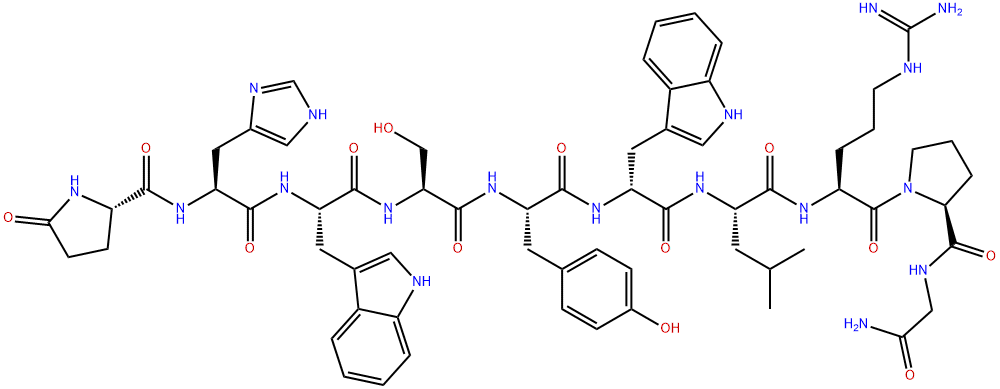
Tesamorelin
- CAS NO.:218949-48-5
- Empirical Formula: C221H366N72O67S
- Molecular Weight: 5135.86
- MDL number: MFCD06199102
- EINECS: 603-809-2
- Update Date: 2026-04-02 17:27:48
What is Tesamorelin?
Description
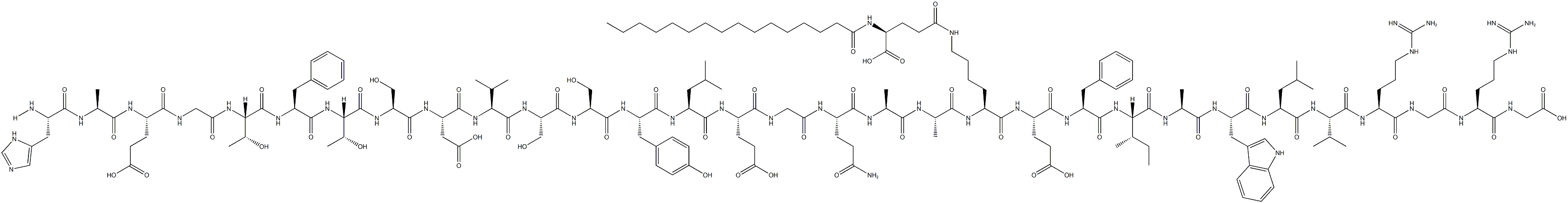
Tesamorelin (usan) consists of the 44 amino acid sequence of human growth hormone releasing factor (GRF), with a 3-hexenoyl group attached to its tyrosine N-terminal residue. It is a new growth hormone releasing factor analogue, which can not only restore normal growth hormone secretion in the body, but also reduce the increased visceral adipose tissue (VAT), improve blood lipid abnormalities and quality of life, and maintain glucose homeostasis in the body.
The Uses of Tesamorelin
Tesamorelin (usan) is indicated for the treatment of excess abdominal fat in HIV-infected patients with lipodystrophy and is not recommended for weight loss.
Definition
FDA Pharm Classes: Tesamorelin is a Growth Hormone Releasing Factor Analog. The physiologic effect of tesamorelin is by means of Increased GHRH Activity.
Side Effects
Common adverse reactions of Tesamorelin (usan) include allergic reactions caused by growth hormone effects (such as arthralgia, peripheral edema, hyperglycemia), such as rash and urticaria, and erythema, itching, pain, urticaria and bleeding at the injection site.
Toxicology
In clinical trials in patients with HIV-associated lipodystrophy, tesamorelin therapy was not associated with de novo elevations in serum enzymes and, in some studies, was associated with decreases in preexisting ALT elevations, possibly mediated by improvements in nonalcoholic fatty liver. Instances of clinically apparent liver injury attributable to tesamorelin use have not been reported.
Properties of Tesamorelin
| storage temp. | 2-8°C Refrigerator |
Safety information for Tesamorelin
Computed Descriptors for Tesamorelin
New Products
Methyl N-((2'-cyano-[1,1'-biphenyl]-4-yl)methyl)-N-nitroso-L-valinate Levofloxacin - Impurity B/ Levofloxacin Related Compound A Ivabradine R isomer Escitalopram - Impurity K Escitalopram Cyno dilol Impurity Paroxetine EP impurity I/Paroxetine Anhydrous EP Impurity I 2-Amino-5-Bromo-6-Chloropyridine 3-Amino-5-bromopicolinic acid 1-(7,8-Dinitro-4,5-dihydro-1H-1,5-methanobenzo[d]azepin-3(2H)-yl)-2,2,2-trifluoroethanone 2-Amino-3-Bromo-5-Cyanopyridine 2-Amino-3-Chloropyridine 2,5-Dibromo-3-MethylpyridineRelated products of tetrahydrofuran
You may like
-
 Tesamorelin 10 Mg Peptide, Packaging Type: Vial, 218949-48-5View Details
Tesamorelin 10 Mg Peptide, Packaging Type: Vial, 218949-48-5View Details
218949-48-5 -
 Tesamorelin 2mg Vial, Powder, Packaging Type: BottleView Details
Tesamorelin 2mg Vial, Powder, Packaging Type: BottleView Details
218949-48-5 -
 Tesamorelin (5mg), 1mg/vialView Details
Tesamorelin (5mg), 1mg/vialView Details
218949-48-5 -
 17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 -
 Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
120279-90-5 -
 76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 -
 118-23-0 NLT 95%View Details
118-23-0 NLT 95%View Details
118-23-0 -
 573704-41-3 NLT 95%View Details
573704-41-3 NLT 95%View Details
573704-41-3