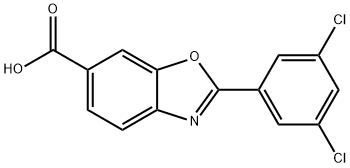
Tafamidis Meglumine
- CAS NO.:951395-08-7
- Empirical Formula: C21H24Cl2N2O8
- Molecular Weight: 503.33
- MDL number: MFCD28502032
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-13 15:00:32

What is Tafamidis Meglumine?
Description
In November 2011, the European Commission approved tafamidis meglumine (Fx-1006A, PF-06291826) for the treatment of transthyretin familial amyloid polyneuropathy (TTR-FAP) in adult patients with stage 1 symptomatic polyneuropathy. Tafamidis stabilizes both the wild type and mutant forms of TTR tetramer and prevents tetramer dissociation by noncooperatively binding to the two thyroxine binding sites. Tafamidis is the first approved medicine for TTR-FAP. The Kd values for tafamidis for the two thyroxine binding sites on TTR, as determined by isothermal titration calorimetry, were 3 nM and 278 nM, respectively. In another in vitro study using wild type TTR, V30M mutant TTR, and V122I mutant TTR, it was shown that tafamidis inhibited fibril formation in a concentration-dependent manner reaching EC50 at a tafamidis:TTR stoichiometry of <1 (EC50 was in the range of 2.7–3.2 μM, corresponding to a tafamidis:TTR stoichiometry range of 0.75–0.9). Tafamidis has been synthesized by coupling 4-amino-3- hydroxybenzoic acid with 3,5-dichlorobenzoyl chloride followed by dehydration using p-toluenesulfonic acid.
Originator
Scripps Research Institute (United States)
The Uses of Tafamidis Meglumine
Tafamidis Meglumine is used as a potential therapeutics for COVID-19 and related viral infections
Definition
ChEBI: Tafamidis meglumine is an organoammonium salt obtained by combining tafamidis with one molar equivalent of 1-deoxy-1-(methylamino)-D-glucitol. Used for the amelioration of transthyretin-related hereditary amyloidosis. It has a role as a central nervous system drug. It contains a tafamidis(1-).
brand name
Vyndaqel
Clinical Use
Tafamidis meglumine is a transthyretin amyloid inhibitor that was approved for the treatment of transthyretin amyloid polyneuropathy (ATTR-PN) and transthyretin familial amyloid polyneropathy (TTR-FAP). These diseases represent a rare autosomal neurodegenerative disorder characterized by autonomic, sensory and motor impairment which are typically fatal. Tafamidis was discovered at The Scripps Research Institute and developed by Pfizer.
Synthesis
Numerous synthetic routes have been reported including the use of direct CH activation to form the key biaryl bond. Although only reported on small scale, the most likely production route is detailed in the scheme.
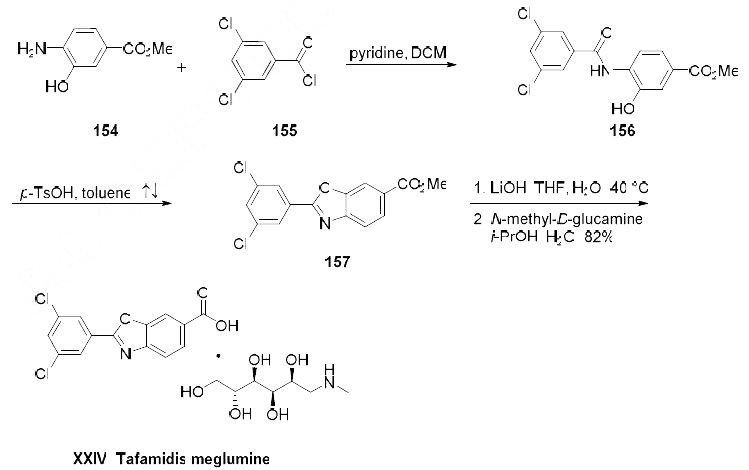
Condensation of methyl 4-amino-3-hydroxybenzoic acid (154) with 3,5-dichlorobenzoyl chloride (155) in refluxing pyridine gave intermediate amide 156 which underwent cycylization upon treatment with p-TsOH in refluxing toluene producing benzoxazole 157. Saponification of the methyl ester with LiOH (aq.) afforded tafamidis. The free acid was treated with N-methyl-D-glutamine to provide tafamidis meglumine (XXIV) in 82% yield.
Properties of Tafamidis Meglumine
| Melting point: | 195 - 198°C |
| storage temp. | -20°C Freezer, Under inert atmosphere |
| solubility | DMSO (Sparingly, Heated, Sonicated) |
| form | Solid |
| color | White to Off-White |
Safety information for Tafamidis Meglumine
| Signal word | Danger |
| Pictogram(s) |
 Health Hazard GHS08 |
| GHS Hazard Statements |
H360:Reproductive toxicity |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P202:Do not handle until all safety precautions have been read and understood. P281:Use personal protective equipment as required. P308+P313:IF exposed or concerned: Get medical advice/attention. P405:Store locked up. P501:Dispose of contents/container to..… |
Computed Descriptors for Tafamidis Meglumine
Tafamidis Meglumine manufacturer
SRINI PHARMACEUTICALS PVT LTD
New Products
Cyclopentane-1,2-dione 2,6-Dibromoaniline 5-Hydroxy-3,9-dimethyl-7H-benzo[c]fluoren-7-one Methyl 4-(acetyloxy)-6-methoxy-1-(4-methoxyphenyl)-2naphthalenecarboxylate 4-Hydroxy-6,7-dimethoxy-1-phenyl-2naphthalenecarboxylic acid (3,5-Dibromophenyl)[4-(trifluoromethyl)phenyl]methanone (4-butoxyphenyl)(4-fluorophenyl)methanone (4-Bromophenyl)(3,4-dimethoxyphenyl)methanone Cefuroxime EP Impurity-A N-Nitroso hydroxy Cetrizine EP Impurity-A Noradrenaline EP Impurity D/Noradrenaline Methyl Ether Cetirizine EP Impurity A/Cetirizine CBHP Impurity Lantanoprost rc B Clidinium Bromide Impurity Benzyl-N,4-dimethylpiperidine-3- amine dihydrochloride 3-(4-phenoxyphenyl)-1H- pyrazolo[3,4- d]pyrimidin-4- amine 4-Ethyl-4-hydroxy-1,4,7,8-tetrahydro-3H,10H-spiro[pyrano[3,4-f]indolizine-6,2'-[1,3]dioxolane]-3,10-dione (E)-N'-(2-cyano-4-nitrophenyl)- N,N-dimethylformimidamide N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide 5-bromopyridin-2-amine 2-Chloro Benzylcyanide 3-chlorobenzyl cyanide 3,4 Diethoxy Benzylcyanide 3,4 Dimethoxy BenzylcyanideRelated products of tetrahydrofuran
You may like
-
 951395-08-7 Tafamidis Meglumine 98%View Details
951395-08-7 Tafamidis Meglumine 98%View Details
951395-08-7 -
 951395-08-7 99%View Details
951395-08-7 99%View Details
951395-08-7 -
 Tafamidis meglumine 98%View Details
Tafamidis meglumine 98%View Details -
 TAFAMIDIS MEGLUMINE 98%View Details
TAFAMIDIS MEGLUMINE 98%View Details -
 Tafamidis meglumine 98%View Details
Tafamidis meglumine 98%View Details -
 951395-08-7 98%View Details
951395-08-7 98%View Details
951395-08-7 -
 Tafamidis meglumine 99%View Details
Tafamidis meglumine 99%View Details
951395-08-7 -
 Tafamidis Meglumine 98%View Details
Tafamidis Meglumine 98%View Details