SalMeterol EP IMpurity D
- CAS NO.:1391052-04-2
- Empirical Formula: C34H47NO7
- Molecular Weight: 581.75
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-04-23 13:02:50
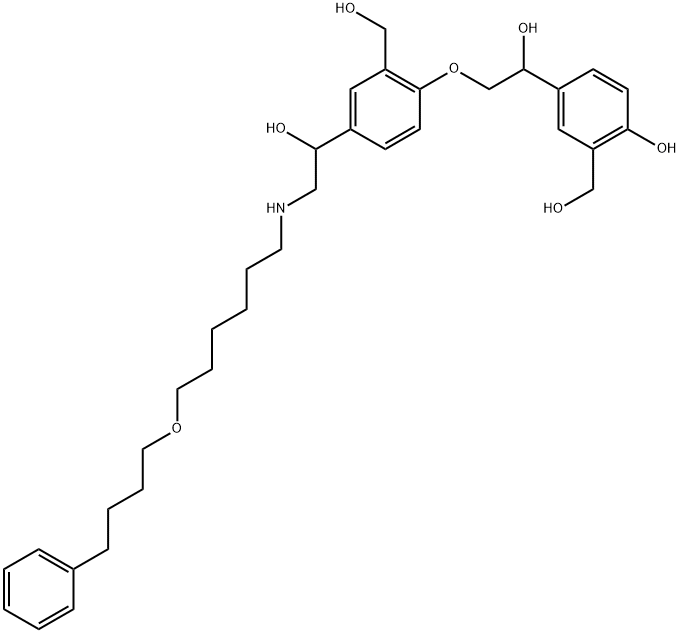
What is SalMeterol EP IMpurity D?
The Uses of SalMeterol EP IMpurity D
4-O-[2-Hydroxy-2-[4-hydroxy-3-(hydroxymethyl)phenyl]ethyl] Salmeterol (Salmeterol EP Impurity D) is an impurity of the β2-Adrenergic agonist Salmeterol (S090100).
The Uses of SalMeterol EP IMpurity D
4-O-[2-Hydroxy-2-[4-hydroxy-3-(hydroxymethyl)phenyl]ethyl] Salmeterol is an impurity of the β2-Adrenergic agonist Salmeterol (S090100).
Properties of SalMeterol EP IMpurity D
| Melting point: | 48-55°C |
| Boiling point: | 784.4±60.0 °C(Predicted) |
| Density | 1.194±0.06 g/cm3(Predicted) |
| storage temp. | Hygroscopic, -20°C Freezer, Under inert atmosphere |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| form | Solid |
| pka | 9.89±0.31(Predicted) |
| color | Orange to Red |
| Stability: | Very Hygroscopic |
Safety information for SalMeterol EP IMpurity D
Computed Descriptors for SalMeterol EP IMpurity D
SalMeterol EP IMpurity D manufacturer
GLP Pharma Standards
3Y
Phone:+91-7893074638
Whatsapp: +91-7893074638
product: Salmeterol EP Impurity D/ Salmeterol O-Alkyl Impurity 1391052-04-2 NLT 95%
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide o-Anisaldehyde 4-Bromobenzaldehyde 3-Fluorobenzaldehyde 2-Chlorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 1-Boc-4-cyanopiperidine ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Vardenafil Bis-sulphonamide(Dimer) Nitroso Irbesartan N-Nitroso Nortriptyline Sulfonic acid Impurity 2-(p-Tolyl)acetaldehyde 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
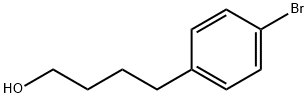
![4-Hydroxy-α1-[[[6-(2-phenylethoxy)hexyl]aMino]Methyl]-1,3-benzenediMethanol](https://img.chemicalbook.in/CAS/GIF/94749-02-7.gif)
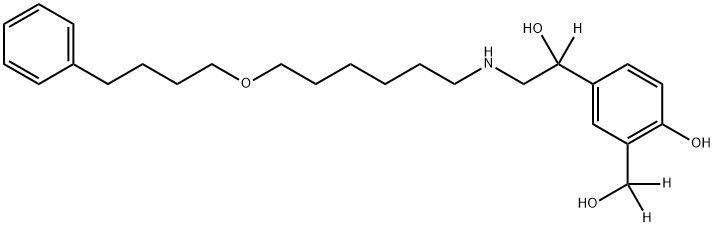
![4-Hydroxy-α1-[[[6-(3-phenylpropoxy)hexyl]aMino]Methyl]-1,3-benzenediMethanol](https://img.chemicalbook.in/CAS/GIF/94749-11-8.gif)
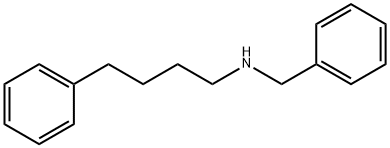
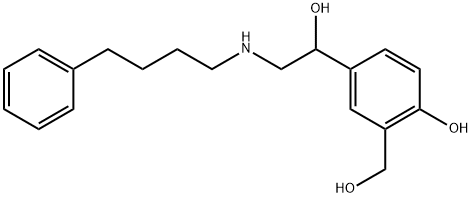
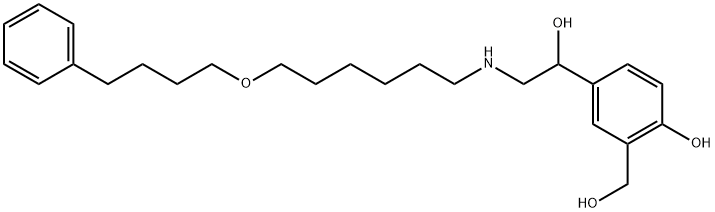
You may like
-
 Salmeterol EP Impurity D 98%View Details
Salmeterol EP Impurity D 98%View Details
1391052-04-2 -
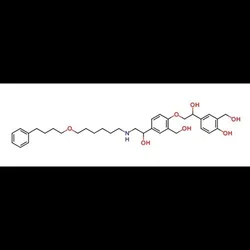 Salmeterol EP Impurity D, Analytical GradeView Details
Salmeterol EP Impurity D, Analytical GradeView Details
1391052-04-2 -
 Salmeterol EP Impurity D/ Salmeterol O-Alkyl Impurity 1391052-04-2 NLT 95%View Details
Salmeterol EP Impurity D/ Salmeterol O-Alkyl Impurity 1391052-04-2 NLT 95%View Details
1391052-04-2 -
 Deuterated Styrene-d8 19361-62-7 97-99%View Details
Deuterated Styrene-d8 19361-62-7 97-99%View Details
19361-62-7 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 (S)-2-Bromobutanoic acid 97-99%View Details
(S)-2-Bromobutanoic acid 97-99%View Details
32659-49-7 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.
