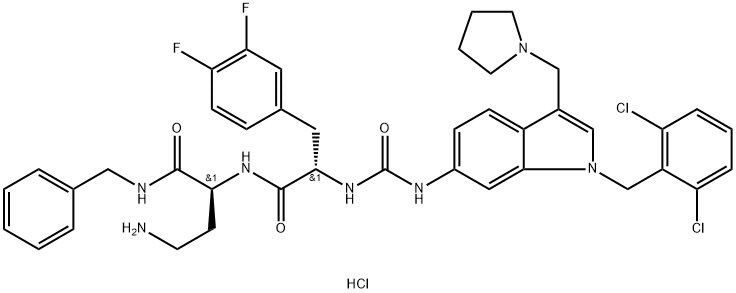
RWJ-56110 dihydrochloride
- CAS NO.:2387505-58-8
- Empirical Formula: C41H44Cl3F2N7O3
- Molecular Weight: 827.2
- MDL number: MFCD22683793
- Update Date: 2025-04-17 18:22:24
What is RWJ-56110 dihydrochloride?
Biological Activity
RWJ-56110 dihydrochloride is a potent, selective, peptide-mimetic inhibitor of PAR-1 activation and internalization (binding IC50=0.44 uM) and shows no effect on PAR-2, PAR-3, or PAR-4. RWJ-56110 dihydrochloride inhibits the aggregation of human platelets induced by both SFLLRN-NH2 (IC50=0.16 μM) and thrombin (IC50=0.34 μM), quite selective relative to U46619 . RWJ-56110 dihydrochloride blocks angiogenesis and blocks the formation of new vessels in vivo. RWJ-56110 dihydrochloride induces cell apoptosis[1][2]. Proteinase-activated receptors (PARs) are a family of G protein-coupled receptors activated by the proteolytic cleavage of their N-terminal extracellular domain, exposing a new amino terminal sequence that functions as a tethered ligand to activate the receptors.RWJ56110 inhibits the aggregation of human platelets induced by both SFLLRN-NH2 (IC50=0.16 μM) and thrombin (IC50=0.34 μM) while being quite selective relative to collagen and the thromboxane mimetic U46619 [1].RWJ-56110 dihydrochloride is fully inhibits thrombin-induced RASMC proliferation with an IC50 value of 3.5 μM. RWJ-56110 dihydrochloride shows blockade of thrombin's action with RASMC calcium mobilization (IC50=0.12 μM), as well as with HMVEC (IC50=0.13 μM) and HASMC calcium mobilization (IC50=0.17 μM)[1].RWJ56110 (0.1-10 μM; 24-96 hours) inhibits endothelial cell growth dose-dependently, with half-maximal inhibitory concentration of RWJ56110 is approximately 10 μM[2].RWJ56110 (0.1-10 μM; 6 hours) inhibits DNA synthesis of endothelial cells in a thymidine incorporation assays. Endothelial cells are in fast-growing state (50-60% confluence), RWJ56110 inhibits cell DNA synthesis in a dose-dependent manner, but when cells that are in the quiescent state (100% confluent), the inhibitory effect of PAR-1 antagonists is much less pronounced[2].RWJ56110 (0.1-10 μM; pretreatment for 15 min) inhibits thrombin-induced Erk1/2 activation in a concentration-dependent manner. However, when endothelial cells are stimulated by FBS (final concentration 4%), it reduces partially the activated levels of Erk1/2[2].RWJ56110 (30 μM; 24 hours) has an inhibitory effect on endothelial cell cycle progression. It reduces the percentage of cells in the S phase, while alterations in the percentages of G1 and G2/M cells are less pronounced[2]. Western Blot Analysis[2] Cell Line: Endothelial cells
Storage
Store at -20°C
References
[1]. Andrade-Gordon, et al.Design, synthesis, and biological characterization of a peptide-mimetic antagonist for a tethered-ligand receptor. oc Natl Acad Sci U S A. 1999 Oct 26;96(22):12257-62. [2]. Panagiota Zania, et al. Blockade of angiogenesis by small molecule antagonists to protease-activated receptor-1: association with endothelial cell growth suppression and induction of apoptosis. J Pharmacol Exp Ther. 2006 Jul;318(1):246-54.
Properties of RWJ-56110 dihydrochloride
| storage temp. | Store at -20°C |
| solubility | Soluble to 25 mM in water and to 100 mM in DMSO |
| form | Solid |
| color | White to light yellow |
Safety information for RWJ-56110 dihydrochloride
Computed Descriptors for RWJ-56110 dihydrochloride
New Products
Pentadecanoic acid 3-Bromophenylacetic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 3′-Hydroxyacetophenone 5-fluoro-1,3-benzodioxole 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 2-Bromo-6-fluoroaniline 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole N-(4-Bromophenyl)-2-chloroacetamide 6-Isopropylpicolinonitrile N-Methyl-3,5-dinitro-2-pyridinamine Phenyl-boronic acid-d5 10,11-Dihdyro-10-oxo-5H-dibenz[b,f]azepine-5-carboxamide-d4 5-(2,4-difluorophenyl)oxazolidin-2-oneYou may like
-
 Deuterated Styrene-d8 97-99%View Details
Deuterated Styrene-d8 97-99%View Details
19361-62-7 -
![1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 -
 32659-49-7 97-99%View Details
32659-49-7 97-99%View Details
32659-49-7 -
 212322-17-3 methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3 methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3
