RESISTOMYCIN
- CAS NO.:20004-62-0
- Empirical Formula: C22H16O6
- Molecular Weight: 376.36
- MDL number: MFCD00597050
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 10:31:43
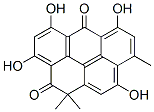
What is RESISTOMYCIN?
The Uses of RESISTOMYCIN
Resistomycin is a tetrahydroxyantrone has been independently isolated by several groups and reported to have a broad range of biological activities. Resistomycin is active against bacteria, including mycobacteria, and viruses. Resistomycin induces apoptosis and inhibits RNA polymerase.
The Uses of RESISTOMYCIN
Resistomycin is a tetrahydroxyanthrone that has been independently isolated by several groups and reported to have a broad range of biological activities. Resistomycin is active against bacteria, including mycobacteria, and viruses. Resistomycin induces apoptosis and inhibits RNA polymerase.
The Uses of RESISTOMYCIN
Resistomycin is an antibiotic RNA polymerase inhibitor and apoptosis inducer.
What are the applications of Application
Resistomycin is an antibiotic RNA polymerase inhibitor and apoptosis inducer
Definition
ChEBI: Resistomycin is a phenanthrol.
Biological Activity
resistomycin is a natural antibiotic.resistomycin is a natural antibiotic produced by several actinobacteriae, such as streptomyces.
in vitro
resistomycin, a tetrahydroxyanthrone derivative, was isolated from the culture broth of streptomyces sulphureus and a similar polyphenolic dianthraquinone, hypericin, was isolated from an extract of hypericum perforatum l. as modulators for apoptosis. in human megakaryoblastic leukemia cmk-7 cells, resistomycin could inhibit apoptosis induced by actinomycin d (ad) with or without acceleration by colcemid (cl), ic50 for inhibition against ad-induced apoptosis was about 0.5 μm and ic50 for inhibition against ad plus cl-induced apoptosis was about 1 μm. cl alone induced weak apoptosis in cells, which could be enhanced by resistomycin. moreover, it was found that both hypericin and resistomycin were able to scavenge superoxide anion radicals at the same rate as luteolin. in addition, pkc in cmk-7 cells could be inhibited by hypericin and luteolin, but not obviously inhibited by resistomycin [1].
References
[1] shiono, y. ,shiono, n.,seo, s., et al. effects of polyphenolic anthrone derivatives, resistomycin and hypercin, on apoptosis in human megakaryoblastic leukemia cmk-7 cell line. z. naturforsch. c. 57(9-10), 923-929 (2002).
Properties of RESISTOMYCIN
| Melting point: | 325℃ (decomposition) |
| Boiling point: | 424.93°C (rough estimate) |
| Density | 1.2465 (rough estimate) |
| refractive index | 1.4450 (estimate) |
| storage temp. | Store at -20°C |
| solubility | Soluble in DMSO;dimethyl formamide |
| form | powder |
| pka | 4.25±0.40(Predicted) |
Safety information for RESISTOMYCIN
Computed Descriptors for RESISTOMYCIN
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 2-Amino-3,5-Dichloro-4-picoline 5-Amino-2-EthoxypyridineYou may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5
