(R)-(-)-IBUPROFEN
- CAS NO.:51146-57-7
- Empirical Formula: C13H18O2
- Molecular Weight: 206.29
- MDL number: MFCD00069290
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-20 15:44:35
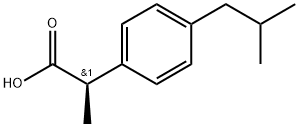
What is (R)-(-)-IBUPROFEN?
Description
Ibuprofen is a non-steroidal anti-inflammatory drug with diverse biochemical actions, most notably inhibiting COX-1 and COX-2 (IC50s = 2.6 and 1.53 μM, respectively). It is commonly synthesized as a racemic mixture of (S) and (R) isomers (Item No. item 70280). (R)-Ibuprofen is an enantiomer that is generally not considered a COX inhibitor and is instead thought to be involved in pathways of lipid metabolism as it is incorporated into triglycerides along with fatty acids. The (R) enantiomer can, however, inhibit NF-κB activation (IC50 = 121.8 μM) in response to T-cell stimulation as well as block superoxide formation, β-glucuronidase release, and LTB4 generation by stimulated neutrophils (IC50 values range from 40-100 μM). 50-60% of (R)-ibuprofen is inverted to (S)-ibuprofen in humans after oral administration.
Chemical properties
White Crystalline Solid
The Uses of (R)-(-)-IBUPROFEN
A nonsteroidal anti-inflammatory drug (NSAID); activity resides primarily in the (S)-isomer
The Uses of (R)-(-)-IBUPROFEN
A nonsteroidal anti-inflammatory drug (NSAID); activity resides primarily in the (S)-isomer.
What are the applications of Application
(R)-Ibuprofen is an inhibitor of Cox-1 and Cox-2
Definition
ChEBI: Levibuprofen is an ibuprofen. It is an enantiomer of a dexibuprofen.
Purification Methods
Crystallise the (+) and (-) acids from EtOH or aqueous EtOH. The racemate which crystallises from pet ether with m 75-77o is sparingly soluble in H2O and has IR (film) 1705 (C=O), 2300—3700 (OH broad)cm-1. It is used as a non-steroidal anti-inflammatory. [Shiori et al. J Org Chem 43 2936 1978, Kaiser et al. J Pharm Sci 65 269 1976, J Pharm Sci 81 221 1992, Freer Acta Cryst (C) 49 1378 1993 for the (S+)-enantiomer.]
Properties of (R)-(-)-IBUPROFEN
| Melting point: | 41-42°C |
| Boiling point: | 305.14°C (rough estimate) |
| Density | 1.0176 (rough estimate) |
| refractive index | 1.5290 (estimate) |
| storage temp. | Sealed in dry,2-8°C |
| solubility | Chloroform (Slightly), Ethanol (Slightly), Methanol (Slightly) |
| form | White crystalline powder. |
| pka | 4.41±0.10(Predicted) |
| color | Colourless to Pale Beige Oil |
| Water Solubility | 369.3mg/L(25 ºC) |
Safety information for (R)-(-)-IBUPROFEN
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for (R)-(-)-IBUPROFEN
(R)-(-)-IBUPROFEN manufacturer
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran

You may like
-
 R)-(-)-IBUPROFENCAS No: 51146-57-7View Details
R)-(-)-IBUPROFENCAS No: 51146-57-7View Details
51146-57-7 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-Picolinic acid N-oxide 824-40-8 98%View Details
2-Picolinic acid N-oxide 824-40-8 98%View Details
824-40-8 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
