Opicapone
- CAS NO.:923287-50-7
- Empirical Formula: C15H10Cl2N4O6
- Molecular Weight: 413.17
- MDL number: MFCD19443745
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-17 13:05:51
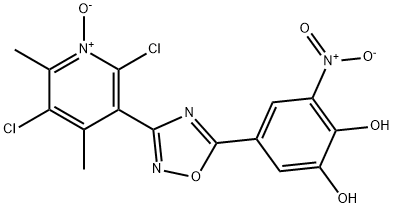
What is Opicapone?
Absorption
Orally administered opicapone demonstrates a linear, dose-dependent absorption profile. Opicapone is rapidly absorbed, with an oral bioavailability of about 20%. Following administration of a single 50 mg dose of opicapone, the median Tmax was two hours, ranging from one to four hours. A moderate fat or moderate calorie meal was shown to decrease the Cmax by 62%, the mean overall plasma exposure (AUC) by 31%, and the Tmax by 4 hours.
Toxicity
There is no reported LD50 data of opicapone. As there is no known antidote for opicapone overdose, overdosage should be managed with symptomatic and supportive treatment. Removal of the drug through gastric lavage and/or inactivation by administering activated charcoal should be considered.
Description
Opicapone is a selective and reversible catechol O-methyltransferase (COMT) inhibitor that was developed by the Portuguese pharmaceutical firm Bial and sold to Neurocrine Biosciences. The drug was approved by the USFDA as adjunctive treatment to levodopa (L-Dopa)/ dopa-decarboxylase inhibitor (DDCI) therapy in adults with Parkinson’s disease (PD) and end-of-dose motor fluctuations that cannot be stabilized on those combinations. In 14- to 15- week double-blind multinational trials and in one-year openlabel extension studies in this patient population, opicapone was an effective and generally well-tolerated adjunctive therapy to L-Dopa plus a DDCI and other PD therapies. During the double-blind phase, adjunctive opicapone (50 mg once daily) provided significantly greater improvements in motor fluctuations than placebo, and no new unexpected safety concerns were identified after treatment with opicapone over a 1.4 year period. Furthermore, no serious cases of hepatotoxicity were reported in clinical trials, which represents a significant safety profile improvement over existing standard-of-care COMT inhibitors enticapone, tolcapone, and nebicapone.
The Uses of Opicapone
Opicapone, is used for the synthesis of novel nitrocatechol-substituted heterocycles, having the ability to inhibit catechol-O-methyltransferase (COMT), used for the treatment of Parkinson`s diseases.
Indications
Opicapone is indicated as adjunctive therapy in adults with Parkinson’s disease and end-of-dose motor fluctuations or “off” episodes whose symptoms cannot be stabilized on the combination therapy of levodopa and DOPA decarboxylase inhibitor (e.g., carbidopa).
Background
Opicapone is a potent, reversible, and peripherally-acting third-generation inhibitor of catechol-o-methyltransferase (COMT), an enzyme involved in the breakdown of various catecholamines including dopamine. Many patients with Parkinson’s disease treated with levodopa plus a dopa decarboxylase (DDC) inhibitor (eg carbidopa) experience motor complications over time, which calls for the management of these symptoms through the use of a dopamine agonist, a monoamine oxidase B inhibitor (selegiline, rasagiline), a catechol-O-methyl transferase (COMT) inhibitor, or amantadine, or using a modified-release formulation of levodopa.
Opicapone is used for adjunct therapy to levodopa and carbidopa in adult patients with Parkinson's disease and end-of-dose motor fluctuations. Opicapone was approved for use by the European Commission in June 2016 and the FDA in April 2020. It is marketed under the brand name Ongentys as once-daily oral capsules. Exhibiting a long duration of action that exceeds 24 hours, opicapone can be administered once-daily and demonstrates the lowest risk for cytotoxicity compared to other catechol-O-methyltransferase inhibitors.
Definition
ChEBI: Opicapone is a ring assembly and an oxadiazole.
Pharmacokinetics
Opicapone is a COMT inhibitor that serves to improve the availability and duration of action of levodopa (L-Dopa), a standard pharmacological treatment for Parkinson's Disease. Opicapone works by blocking the peripheral degradation of L-Dopa mediated by COMT. Opicapone has a long duration of action: following administration of a 50 mg dose, COMT inhibition lasted for more than 24 hours. In clinical trials, opicapone as adjunct therapy to L-Dopa plus a dopa decarboxylase inhibitor significantly improved motor fluctuations than placebo, and the effects were comparable to entacapone.
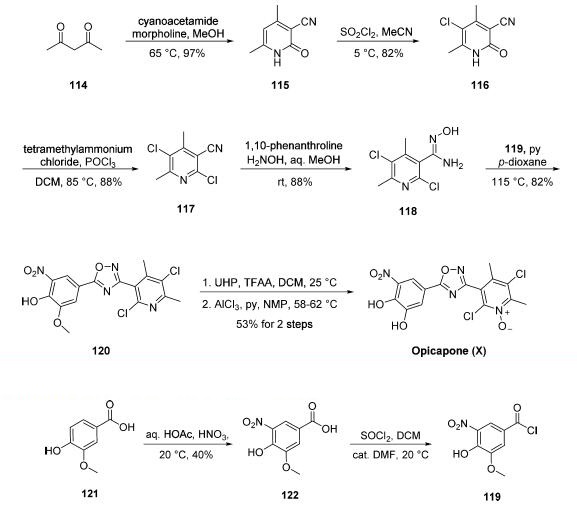
Synthesis
Although several synthetic approaches to opicapone or
opicapone subunits have been disclosed, a synthetic approach
described by Bial was exemplified on a scale capable of
producing 14.4 kg of the active pharmaceutical ingredient
(API). Commercial 2,4-pentanedione (114) was condensed
with cyanoacetamide in warm methanol to give rise to
cyanopyridone 115 in excellent yield. Chlorination
with sulfuryl chloride in chilled acetonitrile followed by
treatment with phosphorus oxychloride resulted in dichloropyridine
117. Next, treatment with hydroxylamine in aqueous
methanol converted nitrile 117 to the corresponding Nhydroxyamidine
118, and this was followed by exposure to
pyridine and acid chloride 119. These operations facilitated a cyclization
reaction, which furnished the key oxadiazole 120 in good yield.
Subjection of 120 to urea hydrogen peroxide (UHP) in
dichloromethane to establish the pyridine N-oxide functionality
within opicapone preceded methyl ether cleavage through the
use of aluminum trichloride in warm pyridine to furnish
opicapone (X) in 53% yield for the two-step sequence.
The preparation of acid chloride 119 involved the nitration
of commercially available benzoic acid 121 followed by thionyl
chloride-mediated conversion of the resulting nitrobenzoic acid
122 to acid chloride 119. Interestingly, although
the nitration step is low-yielding and involves nitric acid, the
authors report an operationally simple isolation method that
has been exemplified on multiple kilogram scale. No yield was
reported for the conversion of 122 to 119.
Metabolism
According to clinical and in vitro studies, sulphation is the primary metabolic pathway of opicapone, forming the inactive metabolite. Opicapone can also undergo glucuronidation, COMT-mediated methylation, reduction, and glutathione conjugation.
As two major circulating metabolites, BIA 9-1103 (3-O-sulphated opicapone) accounts for 67.1% of the total radioactivity and BIA 9-1104 (4-O-methylated opicapone) accounts for 20.5% of the total radioactivity. Other metabolites are generally unquantifiable in plasma samples. Opicapone can undergo N-oxide reduction to form BIA 9-1079, which was shown to be an active metabolite in non-clinical studies; however, it is generally undetectable in humans. Other inactive metabolites include BIA 9-1100, BIA 9-1101, and BIA 9-1106.
Storage
Store at -20°C
Properties of Opicapone
| Boiling point: | 701.1±70.0 °C(Predicted) |
| Density | 1.80±0.1 g/cm3(Predicted) |
| solubility | DMSO : 100 mg/mL (242.03 mM; Need ultrasonic)H2O : < 0.1 mg/mL (insoluble) |
| form | Powder |
| pka | 4.67±0.38(Predicted) |
| color | Light yellow to yellow |
Safety information for Opicapone
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Opicapone
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinYou may like
-
 88660-53-1 >99%View Details
88660-53-1 >99%View Details
88660-53-1 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
