OLANEXIDINE
- CAS NO.:146510-36-3
- Empirical Formula: C17H27Cl2N5
- Molecular Weight: 372.34
- MDL number: MFCD09837698
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-11 16:31:51
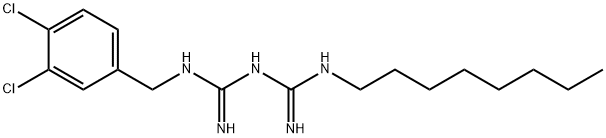
What is OLANEXIDINE?
Description
In July 2015, olanexidine gluconate, a biguanide compound with remarkable antibacterial activity, was approved by the Pharmaceuticals and Medical Devices Agency (PMDA) of Japan for skin antisepsis at surgical sites. The drug was developed and marketed by Otsuka Pharmaceutical in Japan and is available as topical solution (1.5%). Olanexidine gluconate exhibited efficacy against a wide range of bacterial strains, especially Grampositive bacteria. In vitro experiments exploring its mechanism of action indicated that olanexidine interacts with bacterial surface molecules (such as lipopolysaccharides and lipoteichoic acid), disrupting the cell membranes of liposomes. These models suggest that the drug permeates the membranes of both Escherichia coli and Staphylococcus aureus and denatures proteins at relatively high concentrations (>160 g/mL).
Definition
ChEBI: Olanexidine is a dichlorobenzene.
Synthesis
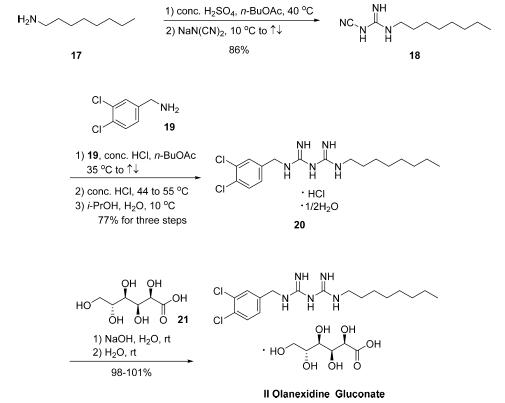
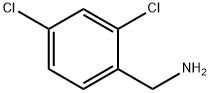
The synthesis of olanexidine gluconate is relatively straightforward, involving the linkage of an n-octyl side chain and a dichlorobenzylamine through a bis-guanidyl lynchpin. The synthesis began with the reaction of commercial noctylamine (17) with sodium dicyanamide in the presence of concentrated sulfuric acid in refluxing n-butyl acetate to give rise to 1-cyano-3-octylguanidine (18) in 86% yield . Conditions employed to subsequently secure biguanidine 20 as the HCl salt hemihydrate in 77% yield were nearly identical to those used for the conversion of 17 to 18. Finally, treatment of 20 with sodium hydroxide in the presence of gluconic acid (21) gave rise to olanexidin gluconate (II) in almost quantitative yield.
Properties of OLANEXIDINE
| Boiling point: | 454.7±55.0 °C(Predicted) |
| Density | 1.22±0.1 g/cm3(Predicted) |
| pka | 11.92±0.10(Predicted) |
Safety information for OLANEXIDINE
Computed Descriptors for OLANEXIDINE
New Products
Pentadecanoic acid 3-Bromophenylacetic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 3′-Hydroxyacetophenone 5-fluoro-1,3-benzodioxole 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 2-Bromo-6-fluoroaniline 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole N-(4-Bromophenyl)-2-chloroacetamide 6-Isopropylpicolinonitrile N-Methyl-3,5-dinitro-2-pyridinamine Phenyl-boronic acid-d5 10,11-Dihdyro-10-oxo-5H-dibenz[b,f]azepine-5-carboxamide-d4 5-(2,4-difluorophenyl)oxazolidin-2-oneRelated products of tetrahydrofuran


You may like
-
 Deuterated Styrene-d8 97-99%View Details
Deuterated Styrene-d8 97-99%View Details
19361-62-7 -
![1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 -
 32659-49-7 97-99%View Details
32659-49-7 97-99%View Details
32659-49-7 -
 212322-17-3 methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3 methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3
