Lithium peroxide
Synonym(s):Dilithium peroxide;Lithium oxide (Li2 O2 )
- CAS NO.:12031-80-0
- Empirical Formula: Li2O2
- Molecular Weight: 45.88
- MDL number: MFCD00016184
- EINECS: 234-758-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-01 16:59:24
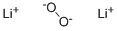
What is Lithium peroxide?
Chemical properties
Lithium peroxide is a white crystalline solid or pale yellow powder which is thermodynamically stable at room temperature. The substance decomposes on heating to 195°C (the exact decomposition temperature is subject to dispute) with the formation of lithium oxide and oxygen. The decomposition forms the basis for one preparation of lithium oxide. High-purity lithium peroxide may be stored for long periods of time with no significant decomposition.
The reaction of carbon dioxide and lithium peroxide is noted above. When lithium peroxide is exposed to the air, lithium carbonate is the final product. Dissolving pure lithium peroxide in water produces an alkaline solution containing lithium ions and hydroperoxide ions. The decomposition of the solution is typical in that oxygen is released on heating or in the presence of a catalyst. The water solubility of lithium peroxide decreases with increasing temperature.
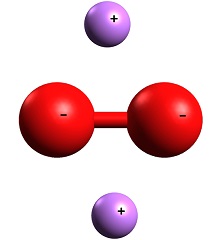
Chemical properties
Lithium peroxide, Li2O2, Mr 45.9, 2.36 g/cm3, has an active oxygen content of 34.8%, which is the highest of all metal peroxides. Lithium peroxide is pale yellow solid, stable at ambient temperature, and not hygroscopic. On heating to about 300 °C, it loses oxygen and forms lithium monoxide.
The Uses of Lithium peroxide
At this time no important industrial uses of lithium peroxide are known. One interesting potential application is in the field of atmosphere regeneration for undersea and space applications, since the compound reacts with carbon dioxide to release oxygen: Li2O2+C02 -> Li2CO3 + 0.5O2.
The Uses of Lithium peroxide
Lithium peroxide is used in air purifiers. It is also used to produce high-purity oxygen. It acts as a catalyst for polymerization of styrene to polystyrene. Further, it serves as a curing agent for special polymers and a source of oxygen in sealed spaces such as submarines and in breathing apparatus.
What are the applications of Application
Lithium peroxide is a chemical used in air purifiers
Preparation
Lithium peroxide is prepared industrially by the reaction of lithium hydroxide monohydrate with hydrogen peroxide which yields lithium hydroperoxide monohydrate.
LiOH·H20 + H202 → LiOOH·H20+H20
The hydroperoxide may be dehydrated by heating in a vacuum to yield the peroxide.
2LiOOH·H20 → Li202+H202 + 2H20
General Description
A white powder or sandy yellow granular solid. Irritates skin, eyes and mucous membranes. Used to produce a supply of high-purity oxygen.
Air & Water Reactions
Contact with water or moist air generates a large amount of heat and corrosive, alkaline lithium hydroxide [AAR 1991].
Reactivity Profile
Lithium peroxide is strongly basic and an extremely powerful oxidizing agent. Accelerates the combustion of other materials, especially organic materials, involved in a fire. Can ignite wood, paper, oil, clothing, etc. on contact. May react explosively with hydrocarbons (fuels). Exposure to heat in a closed container may result in a vigorous reaction that violently ruptures the container.
Health Hazard
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire may produce irritating and/or toxic gases. Toxic fumes or dust may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Runoff from fire control or dilution water may cause pollution.
Fire Hazard
May explode from friction, heat or contamination. These substances will accelerate burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). Some will react explosively with hydrocarbons (fuels). Containers may explode when heated. Runoff may create fire or explosion hazard.
Flammability and Explosibility
Not classified
Safety Profile
A powerful oxidizer and irritant to skin, eyes, and mucous membranes. A very dangerous fire hazard because it is an extremely powerful oxidizing agent. Will react with water or steam to produce heat; on contact with reducing materials, can react vigorously. See also LITHIUM COMPOUNDS, PEROXIDES, and PEROXIDES, INORGANIC.
Properties of Lithium peroxide
| Melting point: | 340°C (dec.) |
| Density | 2.310 |
| solubility | Soluble in anhydrous acetic acid. Insoluble in alcohol. |
| form | Powder |
| color | White to pale yellow |
| Water Solubility | solubility in H2O is 8% (20°C); solubility in acetic acid is 5.6% (20°C); insoluble absolute alcohol (20°C) [HAW93] |
| Sensitive | Air & Moisture Sensitive |
| CAS DataBase Reference | 12031-80-0(CAS DataBase Reference) |
| EPA Substance Registry System | Lithium peroxide (Li2(O2)) (12031-80-0) |
Safety information for Lithium peroxide
| Signal word | Danger |
| Pictogram(s) |
 Flame Over Circle Oxidizers GHS03  Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H272:Oxidising liquids;Oxidising solids H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. |
Computed Descriptors for Lithium peroxide
Lithium peroxide manufacturer
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide o-Anisaldehyde 4-Bromobenzaldehyde 3-Fluorobenzaldehyde 2-Chlorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 1-Boc-4-cyanopiperidine ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Vardenafil Bis-sulphonamide(Dimer) Nitroso Irbesartan N-Nitroso Nortriptyline Sulfonic acid Impurity 2-(p-Tolyl)acetaldehyde 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran

You may like
-
 Lithium peroxide CAS 12031-80-0View Details
Lithium peroxide CAS 12031-80-0View Details
12031-80-0 -
 Lithium peroxide CAS 12031-80-0View Details
Lithium peroxide CAS 12031-80-0View Details
12031-80-0 -
 Lithium peroxide CAS 12031-80-0View Details
Lithium peroxide CAS 12031-80-0View Details
12031-80-0 -
 Industrial Grade Lithium Peroxide PowderView Details
Industrial Grade Lithium Peroxide PowderView Details
6867-30-7 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 (S)-2-Bromobutanoic acid 97-99%View Details
(S)-2-Bromobutanoic acid 97-99%View Details
32659-49-7 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2
