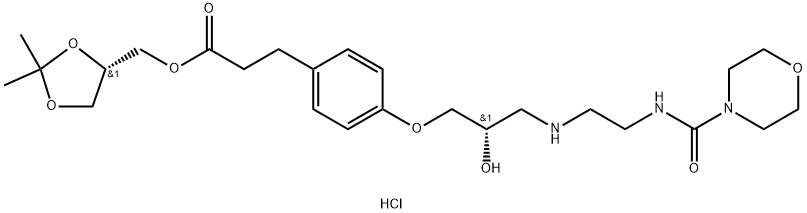
Landiolol
- CAS NO.:133242-30-5
- Empirical Formula: C25H39N3O8
- Molecular Weight: 509.59
- MDL number: MFCD00911043
- EINECS: 1592732-453-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-12 18:10:29

What is Landiolol?
Description
Landiolol was launched as iv infusion for the treatment of tachyarrhythmia during surgery. This structurally related derivative of esmolol can be synthesized in 3 linear steps from 3-(4-hydroxyphenyl)propionic acid by successive esterification followed by alkylation of the phenol function with (2S)-glycidyltosylate and opening of the resulting epoxide by the appropriate amine. Landiolol is an ultra short acting PI-adrenergic blocker more cardioselective (βI/β2 = 255) than esmolol (βi/β2 = 32). It showed 6-8 times greater efficiency compared to esmolol in reducing isoproterenol-induced increase in heart rate and ventricular contraction in anesthetized dogs. In clinical trials, landiolol was effective against a variety of arrhythmias with efficacy seen in patients with atrial fibrillation, proxysmal supraventricular tachycardia, ventricular tachycardia and premature complexes. Landiolol produced a doserelated pharmacokinetic behavior, has a rapid onset of action (10 min.) and is rapidly hydrolyzed to inactive acidic metabolites by esterases after iv administration. This results in an ultra-short half-life (approx. 3 min.) and p-blocade, allowing rapid termination of the drug effect by termination of infusion if side effects occur. Hypertension was the most frequent adverse event and resolved in less than 30 min. after drug withdrawal.
Originator
Ono Pharmaceutical (Japan)
The Uses of Landiolol
Landiolol-D4 is a labelled analogue of Landiolol (L173900).
Definition
ChEBI: Landiolol is a member of morpholines.
brand name
Onoact
Synthesis
Landiolol is prepared by the reaction of 2-(morpholine-4-carboxamido)ethanamino hydrochloride and (S)-(2,2-dimethyl-1,3-dioxolan-4-yl)methyl 3-(4-hydroxyphenyl)propanoate. The steps are as follows:
A suspension of (S)-(2,2-dimethyl-1,3-dioxolan-4-yl)methyl 3-(4-((2R)-3-chloro-2-hydroxypropoxy)phenyl)propanoate prepared according Example 3 (0.50 g, 0.00134 mol) in isopropanol (10 ml) is added with 2-(morpholine-4-carboxamido)ethanamino hydrochloride (18) (1.4 g, 0.00670 mol), heated to 30-35°C and dropwise added with 30% NaOH, keeping pH at 10-11.
The mixture is left under stirring at 35-40°C, monitoring by UPLC.
After completion of the reaction, ethyl acetate (20 ml) and water (20 ml) are added and the phases are separated.
The organic phase is added with water (20 ml) and adjusted to pH 3-4 with hydrochloric acid.
The phases are separated and the resulting aqueous phase is then adjusted to pH 10-11 with sodium hydroxide and re-extracted with ethyl acetate (20 ml).
The solvent is then evaporated off under reduced pressure to obtain 0.38 g (55.6%) of a pale yellow oil which solidifies in time to a pale yellow solid.
Properties of Landiolol
| Boiling point: | 727.5±60.0 °C(Predicted) |
| Density | 1.201±0.06 g/cm3(Predicted) |
| pka | 13.73±0.20(Predicted) |
Safety information for Landiolol
Computed Descriptors for Landiolol
New Products
Cyclopentane-1,2-dione 2,6-Dibromoaniline (4-Bromophenyl)(3-methoxyphenyl)methanone 4,5,7-Trimethyl-2(1H)-quinolinone 4-(6-HYDROXYHEXYLOXY)BENZOIC ACID 4-Hydroxy-6-methyl-1-(4-methylphenyl)-2naphthalenecarboxylic acid 2,6-Piperidinedione, 3-hydroxy-1-[(4-methoxyphenyl)methyl]- NA Cefuroxime EP Impurity-A N-Nitroso hydroxy Cetrizine EP Impurity-A Noradrenaline EP Impurity D/Noradrenaline Methyl Ether Cetirizine EP Impurity A/Cetirizine CBHP Impurity Lantanoprost rc B Clidinium Bromide Impurity 4 - fluoro- 2 - methoxy- 5 - nitroaniline 1-(2-amino-5-hydroxy phenyl)propan-1-one tert-butyl 4-(6-((7-cyclo pentyl -6-(dimethyl carbamoyl) -7H-pyrrolo [2,3-d]pyrimidin-2-yl) amino)pyridin-3-yl) piperazine-1-carboxylate (4-Fluoro-2-methoxy-5-nitro- phenyl)-[4- (1-methyl-1H- indol-3- yl)-pyrimidin-2-yl]- amine (3R,4R)-1-benzyl-N,4-dimethylpiperidin-3- amine 5-bromo-2-chloro-N- cyclopentylpyrimidin-4- amine 2-Chloro Benzylcyanide 3,4 Diethoxy Benzylcyanide 3-chlorobenzyl cyanide 3,4 Dimethoxy BenzylcyanideRelated products of tetrahydrofuran


You may like
-
 4-Hydroxy-6-methyl-1-(4-methylphenyl)-2naphthalenecarboxylic acid 98+View Details
4-Hydroxy-6-methyl-1-(4-methylphenyl)-2naphthalenecarboxylic acid 98+View Details
101894-09-1 -
 83883-25-4 98+View Details
83883-25-4 98+View Details
83883-25-4 -
 54598-15-1 4,5,7-Trimethyl-2(1H)-quinolinone 98+View Details
54598-15-1 4,5,7-Trimethyl-2(1H)-quinolinone 98+View Details
54598-15-1 -
 4-(6-HYDROXYHEXYLOXY)BENZOIC ACID 83883-25-4 98+View Details
4-(6-HYDROXYHEXYLOXY)BENZOIC ACID 83883-25-4 98+View Details
83883-25-4 -
 39973-47-2 NA 98+View Details
39973-47-2 NA 98+View Details
39973-47-2 -
 2357109-89-6 98+View Details
2357109-89-6 98+View Details
2357109-89-6 -
 39973-47-2 NA 98+View Details
39973-47-2 NA 98+View Details
39973-47-2 -
 2357109-89-6 98+View Details
2357109-89-6 98+View Details
2357109-89-6
