L-Milnacipran Hydrochloride
- CAS NO.:96847-54-0
- Empirical Formula: C15H22N2O
- Molecular Weight: 246.35
- MDL number: MFCD12031415
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-04-17 18:22:24
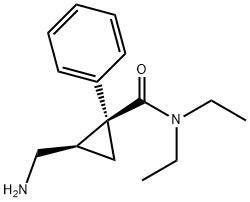
What is L-Milnacipran Hydrochloride?
Absorption
The steady-state concentration of levomilnacipran was dose-proportional when administered at a dose ranging from 25 mg to 300 mg (2.5 times the maximum recommended dosage of levomilnacipran) once daily. After daily dosing of 120 mg levomilnacipran, the mean Cmax value was 341 ng/mL, and the mean steady-state AUC value was 5196 ng x h/mL.
The relative bioavailability of oral levomilnacipran extended-release capsules was 92% when compared to oral solution. The median time to peak concentration (Tmax) of levomilnacipran ranges from six to eight hours after oral administration. Levomilnacipran concentration was not significantly affected when it was administered with food.
Toxicity
The oral LD50 in rats was 238 mg/kg.
There is limited clinical experience with levomilnacipran overdose in humans. In clinical studies, cases of ingestions up to 360 mg daily were reported with none being fatal. As there is no known specific antidote, levomilnacipran overdose should be managed with supportive measures, including close medical supervision and monitoring, with the consideration of possible multiple drug involvement. The high volume of distribution of levomilnacipran suggests that dialysis will not be effective in reducing levomilnacipran plasma concentrations.
Definition
ChEBI: Levomilnacipran is a member of acetamides.
Background
Levomilnacipran is a selective serotonin and norepinephrine reuptake inhibitor (SNRI), although it is a more potent inhibitor of norepinephrine reuptake than serotonin reuptake. Levomilnacipran is the more active 1S,2R-enantiomer in the racemate milnacipran. Once administered, interconversion between levomilnacipran and its stereoisomer does not occur in humans. First approved by the FDA on July 25, 2013, levomilnacipran is used to treat major depressive disorder in adults. While levomilnacipran was previously investigated and proposed as a potential treatment for stroke in Europe, the EMA decided against this use.
Indications
Levomilnacipran is a serotonin and norepinephrine reuptake inhibitor indicated for the treatment of major depressive disorder (MDD) in adults.
Pharmacokinetics
Levomilnacipran is an antidepressant that binds with high affinity to human serotonin (5-HT) and norepinephrine (NE) transporters (Ki = 11 and 91 nM, respectively). It potently inhibits 5-HT and NE reuptake (IC50 = 16 - 19 and 11 nM, respectively). Levomilnacipran does not bind to any other receptors, ion channels, or transporters, including serotonergic (5HT1-7), α- and β adrenergic, muscarinic, or histaminergic receptors and Ca2+, Na+, K+ or Cl- channels to a significant degree. Levomilnacipran did not inhibit monoamine oxidase (MAO). Furthermore, levomilnacipran does not prolong the QTc interval to a clinically relevant extent.
Metabolism
Levomilnacipran undergoes desethylation to form desethyl levomilnacipran (or N-desethyl levomilnacipran) and hydroxylation to form p-hydroxy-levomilnacipran, which are pharmacologically inactive. Both oxidative metabolites can undergo further glucuronidation. Desethylation is primarily catalyzed by CYP3A4 with minor contributions by CYP2C8, 2C19, 2D6, and 2J2.
Properties of L-Milnacipran Hydrochloride
| Boiling point: | 393.0±21.0 °C(Predicted) |
| Density | 1.077±0.06 g/cm3(Predicted) |
| pka | 10.36±0.29(Predicted) |
Safety information for L-Milnacipran Hydrochloride
Computed Descriptors for L-Milnacipran Hydrochloride
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran
You may like
-
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
S-Clopidogrel N-Methyl Impurity 1346605-15-9 NLT 95%View Details
1346605-15-9 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4 -
 Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
Losartan Propyl BCFI 124750-49-8 NLT 95%View Details
124750-49-8
