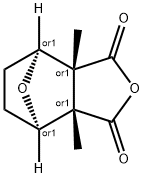
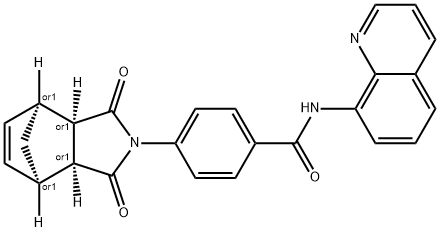
IWR-1-endo
Synonym(s):Inhibitor of Wnt Response-1, TNKS1/2 Inhibitor II, Tankyrase1/2 Inhibitor II, Wnt Pathway Inhibitor I;4-(1,3,3a,4,7,7a-Hexahydro-1,3-dioxo-4,7-methano-2H-isoindol-2-yl)-N-8-quinolinyl-Benzamide;InSolution Wnt Antagonist I, IWR-1-endo - CAS 1127442-82-3 - Calbiochem;IWR1
- CAS NO.:1127442-82-3
- Empirical Formula: C25H19N3O3
- Molecular Weight: 409.44
- MDL number: MFCD18086875
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-18 12:06:49
What is IWR-1-endo?
Description
IWR-1 endo (1127442-82-3) is a potent inhibitor of Wnt signaling (IC50=180 nM).1?Inhibits zebrafish tailfin regeneration (0.5 mM).2?Acts via inhibition of tankyrase and attenuates Wnt/β-catenin signaling in cancer stem-like cells.3?Promotes self-renewal and maintains pluripotency of human embryonic stem cells.4?Promotes differentiation of pluripotent stem cells into cardiomyocytes.5
The Uses of IWR-1-endo
IWR-1-endo act as inhibitors of Wnt response. It appear that IWR compounds induce stabilization of Axin proteins via a direct interaction, which is a part of the b-catenin destruction complex (consists of Apc, Axin, Ck1 and Gsk3b). Such compounds may be used in the treatment of Wnt protein signaling-related diseases and conditions such as cancer, degenerative diseases, type II diabetes and osteopetrosi s.
The Uses of IWR-1-endo
IWR-3 act as inhibitors of Wnt response. It appear that IWR compounds induce stabilization of Axin proteins via a direct interaction, which is a part of the b-catenin destruction complex (consists of Apc, Axin, Ck1 and Gsk3b).Such compounds may be used in the treatment of Wnt protein signaling-related diseases and conditions such as cancer, degenerative diseases, type II diabetes and osteopetrosis.
What are the applications of Application
IWR-1-endo is a potent inhibitor of the Wnt/β-catenin response, able to increase Axin2 protein levels without changing Apc or GSK3β levels.
Definition
ChEBI: IWR-1-endo is a dicarboximide having an endo bridged phthalimide structure, substituted at nitrogen by a 4-(quinolin-8-ylcarbamoyl)benzoyl group. It has a role as an axin stabilizer and a Wnt signalling inhibitor. It is a dicarboximide, a bridged compound, a member of quinolines and a member of benzamides.
General Description
A cell-permeable p-imidobenzamidoquinoline, endo-diastereomer that is shown to inhibit the activity of TNKS1/PARP5a and TNKS2/PARP5b in in vitro auto-PARsylation assays (IC50 = 131 and 56 nM, respectively) and effectively suppress Wnt-stimulated transcription activity in L-Wnt-STF-based reporter assays (IC50 = 180 nM), while exhibiting little activity against PARP1 or PARP2 (IC50 >18.75 μM). Although both IWR-1-endo and XAV939 (Tankyrase1/2 Inhibitor; >Cat. No. 575545) act as reversible Wnt pathway inhibitors and exhibit similar pharmacological effects both in vitro and in vivo, IWR-1-endo exerts its effect via interaction with Axin, while XAV939 binds TNKS directly.
Biochem/physiol Actions
Cell permeable: yes
Storage
Store at RT
References
1) Chen?et al. (2009),?Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer; Nature Chem. Biol.,?5?100 2) Lu?et al.?(2009),?Structure-activity Relationship Studies of Small-Molecule Inhibitors of Wnt Response; Bioorg. Med. Chem. Lett.,?19?3825 3) Martins-Neves?et al.?(2018),?IWR-1, a tankyrase inhibitor, attenuates Wnt/?-catenin signaling in cancer stem-like cells and inhibits in vivo the growth of subcutaneous human osteosarcoma xenograft; Cancer Lett.,?414?1 4) Kim?et al.?(2013),?Modulations of ?-catenin function maintains mouse epiblast stem cell and human embryonic stem cell self-renewal; Nat. Commun., 4?4403 5) Ren?et al.?(2011),?Small Molecule Wnt Inhibitors Enhance the Efficiency of BMP-4-directed Cardiac Differentiation of Human Pluripotent Stem Cells; J. Mol. Cell. Cardiol.,?51?280
Properties of IWR-1-endo
| Boiling point: | 643.9±55.0 °C(Predicted) |
| alpha | -3.0~+3.0°(20℃/D)(c=0.1,CH3CN) |
| Density | 1.425±0.06 g/cm3(Predicted) |
| storage temp. | room temp |
| solubility | DMSO: soluble5mg/mL, clear |
| pka | 11.88±0.43(Predicted) |
| form | Liquid |
| color | white to beige |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
Safety information for IWR-1-endo
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for IWR-1-endo
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran
You may like
-
 IWR-1 CAS 1127442-82-3View Details
IWR-1 CAS 1127442-82-3View Details
1127442-82-3 -
 IWR-1 CAS 1127442-82-3View Details
IWR-1 CAS 1127442-82-3View Details
1127442-82-3 -
 Wnt Antagonist I, IWR-1-endo CASView Details
Wnt Antagonist I, IWR-1-endo CASView Details -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1