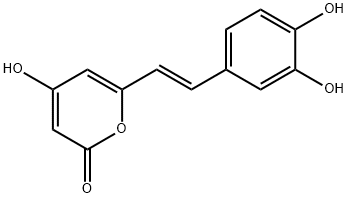
HISPIDIN
Synonym(s):6-(3,4-Dihydroxystyrl)-4-hydroxy-2-pyrone;Hispidin - CAS 555-55-5 - Calbiochem
- CAS NO.:555-55-5
- Empirical Formula: C13H10O5
- Molecular Weight: 246.22
- MDL number: MFCD22199534
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-22 18:57:36
What is HISPIDIN?
Description
Hispidin is a polyphenol originally isolated from P. hispidus that has diverse biological activities, including antioxidant, anti-inflammatory, and cytoprotective properties. In a trolox equivalent antioxidant capacity (TEAC) assay, hispidin scavenges radicals at 14.47 equivalents of trolox . It inhibits transcriptional activity of NF-κB, decreases inducible nitric oxide synthase (iNOS) expression, and decreases the generation of reactive oxygen species (ROS) in LPS-induced macrophage RAW 264.7 cells. Hispidin inhibits apoptosis and increases insulin secretion in hydrogen peroxide-treated RINm5F pancreatic β-cells. It inhibits protein kinase C β (PKCβ; IC50 = 2 μM) with no activity against alkaline phosphatase. Hispidin also inhibits β-secretase (BACE1; IC50 = 4.9 μM) and prolyl endopeptidase (PE; IC50 = 16 μM) but not other serine proteases when used at a concentration of 40 μM (0.6, 0, 8.2, and 3.1% inhibition of chymotrypsin, trypsin, elastase, and tumor necrosis factor-α converting enzyme (TACE), respectively).
The Uses of HISPIDIN
Hispidin is a naturally occuring precursor to fungal luciferin responsible for luminosity in mushrooms.
What are the applications of Application
Hispidin is a cell permeable inhibitor of PKCβ
Definition
ChEBI: Fungal metabolite first found in basidiomycete Inonotus hispidus (formerly Polyporus hispidus).
General Description
Hispidin is a phenolic compound, that is obtained from a medicinal mushroom, Phellinus linteus.
Biochem/physiol Actions
Hispidin exhibits robust antioxidant, anticancer and antidiabetic properties. It has the ability to guard against peroxynitrite-mediated cytotoxicity, DNA damage and the development of hydroxyl radicals.
in vitro
in previous study hispidin was found to reduce cell viability in both mouse and human colon cancer cells, and the apoptotic cell morphological changes were also observed. these results showed accumulation of the sub-g1 cell population and increase in early apoptosis dose-dependently. moreover, hispidin could induce apoptosis via up-regulation of both intrinsic and extrinsic apoptotic pathways. although the molecular mechanism underlying hispidin-induced apoptosis was known to involve the generation of ros, however hispidin was not able to display any apoptosis in the pre-treatment with n-acetyl-l-cysteine, a ros scavenger [1].
in vivo
previous animal study found that the treatment with pkc-activating agent phorbol-12-myristate-13-acetate could attenuate exendin-4-induced relaxations and reduced glp-1r expression in wistar-kyoto rat arteries, which were reversed by hispidin [2].
References
[1] lim jh, lee ym, park sr, kim dh, lim bo. anticancer activity of hispidin via reactive oxygen species-mediated apoptosis in colon cancer cells. anticancer res. 2014 aug;34(8):4087-93.
[2] liu l, liu j, gao y, ng cf, yu x, dou d, huang y. protein kinase cβ mediates downregulated expression of glucagon-like peptide-1 receptor in hypertensive rat renal arteries. j hypertens. 2015 apr;33(4):784-90; discussion 790.
Properties of HISPIDIN
| Melting point: | 312-315℃ |
| Boiling point: | 500.5±50.0 °C(Predicted) |
| Density | 1.657 |
| storage temp. | -20°C |
| solubility | DMSO: >10 mg/mL |
| form | solid |
| pka | 4.90±0.30(Predicted) |
| color | yellow to brown |
| λmax | 369nm(MeOH)(lit.) |
Safety information for HISPIDIN
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H317:Sensitisation, Skin H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for HISPIDIN
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 2-Amino-3,5-Dichloro-4-picoline 5-Amino-2-EthoxypyridineRelated products of tetrahydrofuran
You may like
-
 Hispidin CAS 555-55-5View Details
Hispidin CAS 555-55-5View Details
555-55-5 -
 Hispidin CAS 555-55-5View Details
Hispidin CAS 555-55-5View Details
555-55-5 -
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5