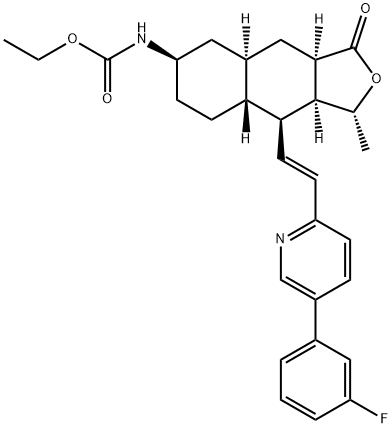
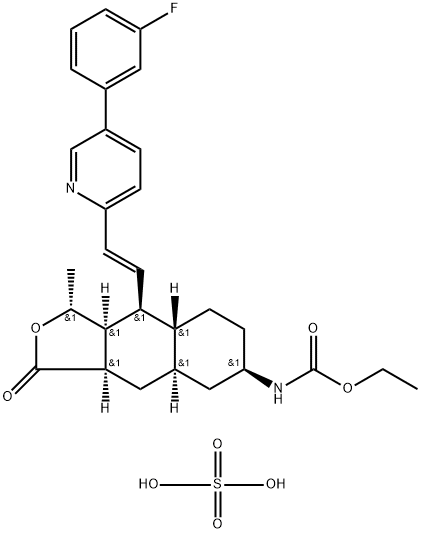
GS9857
- CAS NO.:1535212-07-7
- Empirical Formula: C40H52F4N6O9S
- Molecular Weight: 868.93
- MDL number: MFCD32174311
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 10:31:43
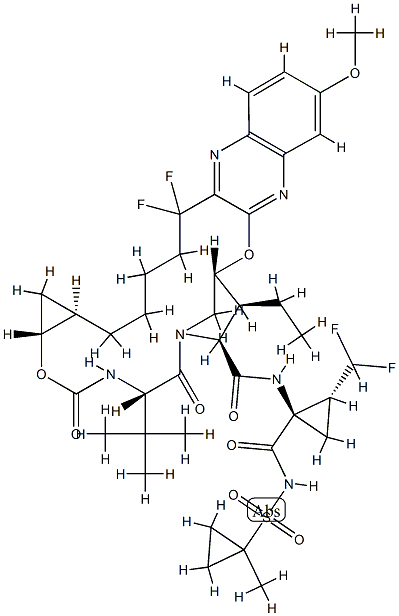
What is GS9857?
Absorption
When provided as the fixed dose combination product Vosevi with Sofosbuvir and Velpatasvir, voxilaprevir reaches a maximum concentration (Cmax) of 192 ng/mL at a maximum time (Tmax) of 4 hours post-dose .
The Uses of GS9857
Voxilaprevir(GS9857) is a hepatitis C virus (HCV) nonstructural (NS) protein 3/4A protease inhibitor (by Gilead) that is used in combination with sofosbuvir and velpatasvir. The combination has the trade name Vosevi and received a positive opinion from the European Committee for Medicinal Products for Human Use in June 2017. On 18 July 2017, Vosevi was approved by the US Food and Drug Administration.
Voxilaprevir is a new chemical entity recently approved in a fixed-dose combination with sofosbuvir1,2 and velpatasvir.3 Like glecaprevir and grazoprevir, voxilaprevir inhibits the NS3/4A protease involved in viral replication. Sofosbuvir is an NS5B nucleotide polymerase inhibitor and velpatasvir is an NS5A inhibitor.
Indications
Vosevi (Voxilaprevir/Sofosbuvir/Velpatasvir) is approved for use in patients with genotypes 1-6 who have been previously treated with an NS5A inhibitor, or patients with genotypes 1a or 3 infection who have previously been treated with an HCV regimen containing Sofosbuvir without an NS5A inhibitor .
Background
Voxilaprevir is a Direct-Acting Antiviral (DAA) medication used as part of combination therapy to treat chronic Hepatitis C, an infectious liver disease caused by infection with Hepatitis C Virus (HCV). HCV is a single-stranded RNA virus that is categorized into nine distinct genotypes, with genotype 1 being the most common in the United States, and affecting 72% of all chronic HCV patients .
Voxilaprevir exerts its antiviral action by reversibly binding and inhibiting the NS3/4A serine protease of Hepatitis C Virus (HCV) . Following viral replication of HCV genetic material and translation into a single polypeptide, Nonstructural Protein 3 (NS3) and its activating cofactor Nonstructural Protein 4A (NS4A) are responsible for cleaving genetic material into the following structural and nonstructural proteins required for assembly into mature virus: NS3, NS4A, NS4B, NS5A, and NS5B . By inhibiting viral protease NS3/4A, voxilaprevir therefore prevents viral replication and function. Treatment options for chronic Hepatitis C have advanced significantly since 2011, with the development of Direct Acting Antivirals (DAAs) such as voxilaprevir.
Voxilaprevir has been available since July 2017 in a fixed dose combination product with sofosbuvir and velpatasvir as the commercially available product Vosevi. Vosevi is approved for the treatment of adult patients with chronic HCV infection with genotype 1, 2, 3, 4, 5, or 6 infection . Notably, Vosevi is approved for use in patients with genotypes 1-6 who have been previously treated with an NS5A inhibitor, or patients with genotypes 1a or 3 infection and have previously been treated with an HCV regimen containing sofosbuvir without an NS5A inhibitor . Prior to Vosevi, there were no approved retreatment options for patients who have previously received, and failed, a regimen containing an NS5A inhibitor for treatment of chronic HCV infection.
What are the applications of Application
Voxilaprevir is a Direct-Acting Antiviral (DAA) medication used as part of combination therapy to treat chronic Hepatitis C, an infectious liver disease caused by infection with Hepatitis C Virus (HCV). HCV is a single-stranded RNA virus that is categorized into nine distinct genotypes, with genotype 1 being the most common in the United States, and affecting 72% of all chronic HCV patients. Voxilaprevir exerts its antiviral action by reversibly binding and inhibiting the NS3/4A serine protease of Hepatitis C Virus (HCV). Following viral replication of HCV genetic material and translation into a single polypeptide, Nonstructural Protein 3 (NS3) and its activating cofactor Nonstructural Protein 4A (NS4A) are responsible for cleaving genetic material into the following structural and nonstructural proteins required for assembly into mature virus: NS3, NS4A, NS4B, NS5A, and NS5B. By inhibiting viral protease NS3/4A, voxilaprevir therefore prevents viral replication and function. Treatment options for chronic Hepatitis C have advanced significantly since 2011, with the development of Direct Acting Antivirals (DAAs) such as voxilaprevir. Voxilaprevir has been available since July 2017 in a fixed dose combination product with [sofosbuvir] and [velpatasvir] as the commercially available product Vosevi. Vosevi is approved for the treatment of adult patients with chronic HCV infection with genotype 1, 2, 3, 4, 5, or 6 infection. Notably, Vosevi is approved for use in patients with genotypes 1-6 who have been previously treated with an NS5A inhibitor, or patients with genotypes 1a or 3 infection and have previously been treated with an HCV regimen containing sofosbuvir without an NS5A inhibitor. Prior to Vosevi, there were no approved retreatment options for patients who have previously received, and failed, a regimen containing an NS5A inhibitor for treatment of chronic HCV infection.
Pharmacokinetics
Voxilaprevir is a direct-acting antiviral agent that targets viral NS3/4A protein and causes a decrease in serum HCV RNA levels. It disrupts HCV replication by specifically inhibiting the critical functions of NS3/4A protein in the replication complex. It does not appear to prolong the QT interval even when given at 9 times the maximum recommended dose .
Metabolism
Voxilaprevir is primarily metabolized by Cytochrome P450 3A4 (CYP3A4) and to a lesser extent by CYP2C8 and CYP1A2.
Properties of GS9857
| Density | 1.42±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMF:30.0(Max Conc. mg/mL);34.52(Max Conc. mM) DMSO:30.0(Max Conc. mg/mL);34.52(Max Conc. mM) Ethanol:1.0(Max Conc. mg/mL);1.15(Max Conc. mM) |
| form | A solid |
| pka | 4.46±0.40(Predicted) |
| color | White to off-white |
Safety information for GS9857
Computed Descriptors for GS9857
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 2-Amino-3,5-Dichloro-4-picoline 5-Amino-2-EthoxypyridineRelated products of tetrahydrofuran
You may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5