dolasetron
- CAS NO.:115956-12-2
- Empirical Formula: C19H20N2O3
- Molecular Weight: 324.37
- MDL number: MFCD00865542
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-22 18:57:36
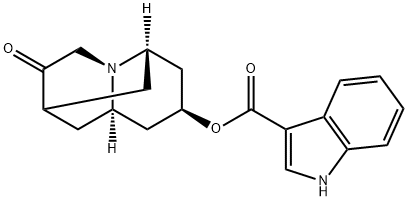
What is dolasetron?
Absorption
Orally-administered dolasetron is well absorbed
Description
Dolasetron is an antagonist of the serotonin (5-HT) receptor subtype 5-HT3 (Ki = 20 nM). It is selective for 5-HT3 receptors over 5-HT1A, 5-HT1B, 5-HT2, dopamine D2, α1-, α2-, β-adrenergic, M1-5 muscarinic acetylcholine, and neurokinin-1 (NK1) receptors (IC50s = >10 μM for all). Dolasetron inhibits 5-HT-induced membrane currents in NG 108-15 cells (IC50 = 3.8 nM). It increases the latency to emesis and reduces the number of vomiting and retching episodes induced by cisplatin in ferrets when administered at doses of 0.5 or 2 mg/kg. Formulations containing dolasetron have been used in the prevention of postoperative or chemotherapy-induced nausea.
Description
Dolasetron, available commercially as Anzement, is an anti-emetic drug that is especially useful to chemotherapy patients. Its mode of action is as a serotonin 5-HT3?receptor antagonist. A new, more scalable synthetic route to dolasteron was featured in a?recent Patent Watch report.
Chemical properties
White Solid
The Uses of dolasetron
Dolasetron is a bridged pseudopelletierine derivative; specific serotonin (5HT3) receptor antagonist.It is used as antiemetic.
The Uses of dolasetron
prevention and treatment of postoperative nausea and vomiting
The Uses of dolasetron
Bridged pseudopelletierine derivative; specific serotonin (5HT3) receptor antagonist. Antiemetic.
Background
Dolasetron is an antinauseant and antiemetic agent indicated for the prevention of nausea and vomiting associated with moderately-emetogenic cancer chemotherapy and for the prevention of postoperative nausea and vomiting. Dolasetron is a highly specific and selective serotonin 5-HT3 receptor antagonist. This drug is not shown to have activity at other known serotonin receptors, and has low affinity for dopamine receptors.
Indications
For the prevention of nausea and vomiting associated with emetogenic cancer chemotherapy, including initial and repeat courses of chemotherapy. Also used for the prevention of postoperative nausea and vomiting. This drug can be used intravenously for the treatment of postoperative nausea and vomiting.
What are the applications of Application
Dolasetron is an antiserotonin compound
Definition
ChEBI: LSM-5418 is an indolyl carboxylic acid.
brand name
Anzemet (Sanofi Aventis.
Pharmacokinetics
Dolasetron is a highly specific and selective serotonin 5-HT3 receptor antagonist, not shown to have activity at other known serotonin receptors and with low affinity for dopamine receptors. It is structurally and pharmacologically related to other 5-HT3 receptor agonists. The serontonin 5-HT3 receptors are located on the nerve terminals of the vagus in the periphery, and centrally in the chemoreceptor trigger zone of the area postrema. It is suggested that chemotherapeutic agents release serotonin from the enterochromaffin cells of the small intestine by causing degenerative changes in the GI tract. The serotonin then stimulates the vagal and splanchnic nerve receptors that project to the medullary vomiting center, as well as the 5-HT3 receptors in the area postrema, thus initiating the vomiting reflex, causing nausea and vomiting.
Metabolism
Hepatic
Properties of dolasetron
| Melting point: | 278 ºC |
| Boiling point: | 535.1±50.0 °C(Predicted) |
| Density | 1.37±0.1 g/cm3(Predicted) |
| storage temp. | Sealed in dry,2-8°C |
| solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Sparingly) |
| form | Solid |
| pka | 15.36±0.30(Predicted) |
| color | White |
| Water Solubility | Soluble |
| CAS DataBase Reference | 115956-12-2 |
Safety information for dolasetron
Computed Descriptors for dolasetron
New Products
2-(2-Ethoxyphenoxy)ethyl bromide 1,3-Dibromo-2,2-dimethoxypropane 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid 8-Bromo-3-methyl xanthine Maleic hydrazide o-Anisaldehyde 4-Bromobenzaldehyde 3-Fluorobenzaldehyde 2-Chlorobenzaldehyde 3-Amino-3-(3-fluorophenyl)propanoic acid 1-Propyl-4-piperidone 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2-Fluoro-6-iodobenzoic acid 3-Pyridineacrylic acid 1-Boc-4-cyanopiperidine ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate Vardenafil Bis-sulphonamide(Dimer) Nitroso Irbesartan N-Nitroso Nortriptyline Sulfonic acid Impurity 2-(p-Tolyl)acetaldehyde 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran



You may like
-
 Deuterated Styrene-d8 19361-62-7 97-99%View Details
Deuterated Styrene-d8 19361-62-7 97-99%View Details
19361-62-7 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 (S)-2-Bromobutanoic acid 97-99%View Details
(S)-2-Bromobutanoic acid 97-99%View Details
32659-49-7 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 methyl 3-amino-4-formylbenzoate 97-99%View Details
methyl 3-amino-4-formylbenzoate 97-99%View Details
212322-17-3 -
![3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
3-bromo-[1,1'-biphenyl]-2-amine 1620885-59-7 97-99%View Details
1620885-59-7 -
![(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) (13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
(13S,17S)-13-methyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate 2590-41-2 97-99%View Details
2590-41-2 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2
