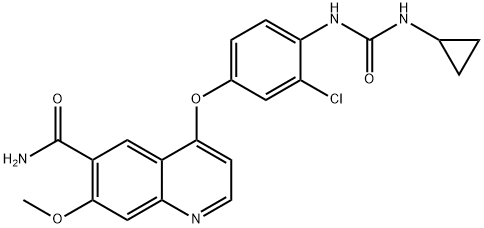
Derazantinib
- CAS NO.:1234356-69-4
- Empirical Formula: C29H29FN4O
- Molecular Weight: 468.57
- MDL number: MFCD30532770
- Update Date: 2026-05-28 03:57:12
What is Derazantinib?
The Uses of Derazantinib
Derazantinib is an intermediate in the preparation of imidazopyridinyl compounds for treating proliferative disorders like cancer, Proteus syndrome and others.
Properties of Derazantinib
| Boiling point: | 615.1±65.0 °C(Predicted) |
| Density | 1.218±0.06 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMSO:47.67(Max Conc. mg/mL);101.73(Max Conc. mM) DMF:30.0(Max Conc. mg/mL);64.02(Max Conc. mM) DMF:PBS (pH 7.2) (1:3):0.25(Max Conc. mg/mL);0.53(Max Conc. mM) |
| form | A crystalline solid |
| pka | 9.04±0.19(Predicted) |
| color | White to yellow |
Safety information for Derazantinib
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P332+P313:IF SKIN irritation occurs: Get medical advice/attention. P337+P313:IF eye irritation persists: Get medical advice/attention. |
Computed Descriptors for Derazantinib
New Products
3-Hydroxypropionitrile 5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -4-(trifluoromethyl)pyridin-2-amine 1,3-Dichloroacetone Ethylene Ketal 4-N-BOC-AMINO-4-CARBOXYTETRAHYDROPYRAN AMino-(3-Methylphenyl)-acetic acid 1-N-Boc-3-(aminoethyl)azetidine 2-Bromo-5-Chloropyridine 2-Picolinic acid N-oxide 4-Butyl Resorcinol 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 5-Amino-2-Iodopyridine 5-Bromo-2-Hydroxy-3-Nitro Pyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 2-Hydroxy-5-methoxybenzoic acid 2-Cyanopyrimidine Xanthene-9-carboxylic acid 4,4-Diethoxybutanenitrile Methyl 4-amino-2,3-difluoro-5-nitrobenzoate 5-fluoro-1,3-benzodioxole 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-(Chloromethyl) quinazolin-4(3H)-one 4-Chloro-2-methyl quinoline 2-Amino-4-phenyl-thiazoleRelated products of tetrahydrofuran
You may like
-
 172843-97-9 4-N-BOC-AMINO-4-CARBOXYTETRAHYDROPYRAN 98+View Details
172843-97-9 4-N-BOC-AMINO-4-CARBOXYTETRAHYDROPYRAN 98+View Details
172843-97-9 -
 944401-57-4 98+View Details
944401-57-4 98+View Details
944401-57-4 -
 1,3-Dichloroacetone Ethylene Ketal 26271-50-1 98+View Details
1,3-Dichloroacetone Ethylene Ketal 26271-50-1 98+View Details
26271-50-1 -
 172843-97-9 4-N-BOC-AMINO-4-CARBOXYTETRAHYDROPYRAN 98+View Details
172843-97-9 4-N-BOC-AMINO-4-CARBOXYTETRAHYDROPYRAN 98+View Details
172843-97-9 -
 944401-57-4 98+View Details
944401-57-4 98+View Details
944401-57-4 -
 AMino-(3-Methylphenyl)-acetic acid 98+View Details
AMino-(3-Methylphenyl)-acetic acid 98+View Details
53519-82-7 -
 1,3-Dichloroacetone Ethylene Ketal 26271-50-1 98+View Details
1,3-Dichloroacetone Ethylene Ketal 26271-50-1 98+View Details
26271-50-1 -
 898271-20-0 1-N-Boc-3-(aminoethyl)azetidine 98+View Details
898271-20-0 1-N-Boc-3-(aminoethyl)azetidine 98+View Details
898271-20-0
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.