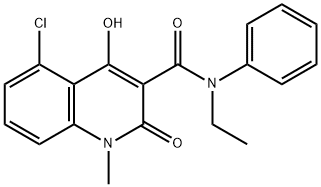
CIVENTICHEM CV-4057
- CAS NO.:248281-84-7
- Empirical Formula: C19H17ClN2O3
- Molecular Weight: 356.8
- MDL number: MFCD08689859
- EINECS: 692-155-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-28 05:03:27
What is CIVENTICHEM CV-4057?
Description
Laquinimod (248281-84-7) is an immunomodulator1 that inhibits inflammation2 and autoimmunity3,4 via activation of the aryl hydrocarbon receptor (AhR)5. It has been investigated as a treatment for multiple sclerosis6 and has shown efficacy in Huntington’s disease models7,8.
Definition
ChEBI: Laquinimod is an aromatic amide.
in vitro
abr-215062 was shown to completely inhibit the development of murine acute experimental autoimmune encephalomyelitis (eae) [1].
in vivo
abr-215062 dose-dependently inhibited disease and showed better disease inhibitory effects as compared to roquinimex (linomide). furthermore, abr-215062 inhibited the inflammation of both cd4+ t cells and macrophages into central nervous tissues [2].
References
J?nsson et al. (2004), Synthesis and Biological Evaluation of New 1,2-Dihydro-4-hydroxy-2-oxo-3-quinolinecarboxamides for Treatment of Autoimmune Disorders: Structure-Activity Relationship; Med. Chem. 47 2075 Rothhammer et al. (2021); Aryl Hydrocarbon Receptor Activation in Astrocytes by Laquinimod Ameliorates Autoimmune Inflammation in the CNS, Neurol. Neuroimmunol. Neuroinflamm., 8 e946 Pitarokoili et al. (2014), Laquinimod exerts strong clinical and immunomodulatory effects in Lewis rat experimental autoimmune neuritis; Neuroimmunol., 274 38 Ott et al. (2019), Laquinimod, a prototypic quinoline-3-carboxamide and aryl hydrocarbon receptor agonist, utilizes CD155-mediated natural killer/dendritic cell interaction to suppress CNS autoimmunity; Neuroinflammation, 16 49 Kaye et al. (2016), Laquinimod arrests experimental autoimmune encephalitis by activating the aryl hydrocarbon receptor; Natl. Acad. Sci. USA., 113 E6145 Th?ne and Linker (2016), Laquinimod in the treatment of multiple sclerosis: a review of the data so far; Drug Des. Devel. Ther., 10 1111 Dobson et al. (2016), Laquinimod dampens hyperactive cytokine production in Huntington’s disease patient myeloid cells; Neurochem., 137 782 Garcia-Miralles et al. (2016), Laquinimod rescues striatal, cortical and white matter pathology and results in modest behavioural improvements in the YAC128 model of Huntington disease; Rep., 6 31652
Properties of CIVENTICHEM CV-4057
| Melting point: | 201 °C (decomp) |
| Boiling point: | 484.8±45.0 °C(Predicted) |
| Density | 1.395±0.06 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | Soluble in DMSO (>25 mg/ml) |
| form | solid |
| pka | 4.50±1.00(Predicted) |
| color | Off-white |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
Safety information for CIVENTICHEM CV-4057
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H301:Acute toxicity,oral |
| Precautionary Statement Codes |
P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. |
Computed Descriptors for CIVENTICHEM CV-4057
New Products
Pentadecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetrahydropyranyl-4-acetic acid Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 5-fluoro-1,3-benzodioxole 2-Bromo-6-fluoroaniline 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran

You may like
-
 Laquinimod 95% CAS 248281-84-7View Details
Laquinimod 95% CAS 248281-84-7View Details
248281-84-7 -
 5-Chloro-N-Et-4-hydroxy-1-methyl-2-oxo-N-Ph-1,2-dihydroquinoline-3-carboxamide CAS 248281-84-7View Details
5-Chloro-N-Et-4-hydroxy-1-methyl-2-oxo-N-Ph-1,2-dihydroquinoline-3-carboxamide CAS 248281-84-7View Details
248281-84-7 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
