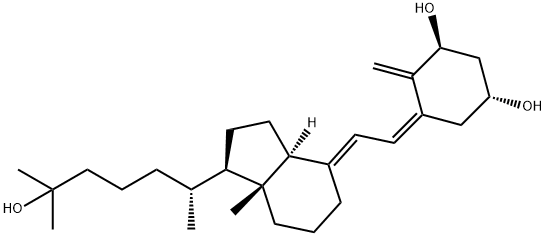
Cholecalciferol Impurity A
- CAS NO.:22350-41-0
- Empirical Formula: C27H44O
- Molecular Weight: 384.64
- MDL number: MFCD00078131
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-13 23:13:44
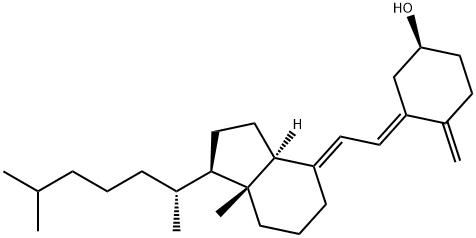
What is Cholecalciferol Impurity A?
Chemical properties
White to Off-White Solid
The Uses of Cholecalciferol Impurity A
5,6-trans-Vitamin D3 is the major photoisomer of Vitamin D3 analog, as an impurity.
Definition
ChEBI: 5,6-trans-vitamin D3 is a member of the class of D3 vitamins that is calciol in which the double bond at position 5 adopts a trans-configuration. During exposure to sunlight, previtamin D3 and vitamin D3 in the skin become photoisomerized to 5,6-trans-vitamin D3. It has a role as a human metabolite. It is a member of D3 vitamins, a seco-cholestane, a secondary alcohol and a hydroxy seco-steroid. It is functionally related to a calciol.
Properties of Cholecalciferol Impurity A
| Boiling point: | 496.4±24.0 °C(Predicted) |
| Density | 0.96±0.1 g/cm3(Predicted) |
| solubility | DMF: 25 mg/ml,DMSO: 3 mg/ml,Ethanol: 30 mg/ml |
| pka | 14.74±0.20(Predicted) |
| form | A crystalline solid |
| color | White to light yellow |
| Stability: | Light Sensitive, Temperature Sensitive |
Safety information for Cholecalciferol Impurity A
Computed Descriptors for Cholecalciferol Impurity A
New Products
2-(2-aminoethyl) benzenesulfonamide Gemcitabine 3-benzoate impurity Dihydroxyphenylacetone Ornidazole EP impurity D Acetyl amino diol impurity Valsartan Benzyl Ester Impurity B N,N'-Dicyclohexylcarbodiimide 3-Bromo-2-Methyl-5-Nitropyridine 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 2-Amino-4-Methylpyridine 4-Aminopyridine 4-dimethylaminopyridineRelated products of tetrahydrofuran


You may like
-
 (5E,7E)-9,10-Secocholesta-5,7,10(19)-trien-3beta-ol CAS 22350-41-0View Details
(5E,7E)-9,10-Secocholesta-5,7,10(19)-trien-3beta-ol CAS 22350-41-0View Details
22350-41-0 -
 Vitamin D3 Injection (Canrise D3 by Healing Pharma), 5 mlView Details
Vitamin D3 Injection (Canrise D3 by Healing Pharma), 5 mlView Details
22350-41-0 -
 4-(Methylnitrosoamino)-butanal NLT 95%View Details
4-(Methylnitrosoamino)-butanal NLT 95%View Details
339362-86-6 -
 848696-99-1 NLT 95%View Details
848696-99-1 NLT 95%View Details
848696-99-1 -
 Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
Ornidazole EP impurity A 1384752-15-1 NLT 95%View Details
1384752-15-1 -
 615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 Diisopropyl oxalate NLT 95%View Details
615-81-6 -
 175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 Lacosamide EP Impurity F NLT 95%View Details
175481-38-6 -
 Valsartan Related Compound A NLT 95%View Details
Valsartan Related Compound A NLT 95%View Details
137862-87-4
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.
