Butylated hydroxytoluene (BHT)
Synonym(s):BHT;Butylated hydroxytoluene;2,6-Di-tert-butyl-4-methylphenol;Butylhydroxytoluene;DBPC
- CAS NO.:128-37-0
- Empirical Formula: C15H24O
- Molecular Weight: 220.35
- MDL number: MFCD00011644
- EINECS: 204-881-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-06-05 18:44:30
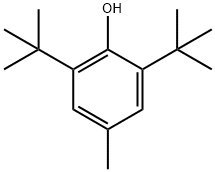
What is Butylated hydroxytoluene (BHT) ?
Description
The antioxidant butylated hydroxytoluene(BHT) is contained in food, adhesive glues, industrial oils and greases, including cutting fluids. Sensitization seems very rare. Formulations containing BHT have been used as antioxidant cosmetic and food additives.
Chemical properties
Butylated hydroxytoluene occurs as a white or pale yellow crystalline solid or powder with a faint characteristic phenolic odor.
Occurrence
Not reported found naturally.
The Uses of Butylated hydroxytoluene (BHT)
Butylated Hydroxytoluene is also known as butylated hydroxy toluene. It is an anti-oxidant that also has preservative and masking capabilities.
Definition
ChEBI: A member of the class of phenols that is 4-methylphenol substituted by tert-butyl groups at positions 2 and 6.
Production Methods
Prepared by the reaction of p-cresol with isobutene.
Preparation
Butylated Hydroxytoluene is produced commercially by the alkylation of para-cresol with isobutylene. Butylated Hydroxytoluene is also produced by several western European manufacturers, production/processing plants in Germany, France, the Netherlands, United Kingdom and Spain.
General Description
White crystalline solid.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Phenols, such as 2,6-Di-tert-butyl-4-methylphenol, do not behave as organic alcohols, as one might guess from the presence of a hydroxyl (-OH) group in their structure. Instead, they react as weak organic acids. Phenols and cresols are much weaker as acids than common carboxylic acids (phenol has Ka = 1.3 x 10^[-10]). These materials are incompatible with strong reducing substances such as hydrides, nitrides, alkali metals, and sulfides. Flammable gas (H2) is often generated, and the heat of the reaction may ignite the gas. Heat is also generated by the acid-base reaction between phenols and bases. Such heating may initiate polymerization of the organic compound. Phenols are sulfonated very readily (for example, by concentrated sulfuric acid at room temperature). The reactions generate heat. Phenols are also nitrated very rapidly, even by dilute nitric acid. Nitrated phenols often explode when heated. Many of them form metal salts that tend toward detonation by rather mild shock. May react with oxidizing materials.
Health Hazard
2,6-Di-tert-butyl-p-cresol or Butylated Hydroxytoluene is of relatively low acute toxicity in animals, and there is no evidence of either acute or chronic effects among exposed workers.
Fire Hazard
2,6-Di-tert-butyl-4-methylphenol is combustible.
Flammability and Explosibility
Non flammable
Pharmaceutical Applications
Butylated hydroxytoluene is used as an antioxidant in
cosmetics, foods, and pharmaceuticals. It is mainly used to
delay or prevent the oxidative rancidity of fats and oils and to
prevent loss of activity of oil-soluble vitamins.
Butylated hydroxytoluene is also used at 0.5–1.0% w/w
concentration in natural or synthetic rubber to provide enhanced
color stability.
Butylated hydroxytoluene has some antiviral activity and has
been used therapeutically to treat herpes simplex labialis.
Contact allergens
This antioxidant is contained in food, adhesive glues, industrial oils, and greases, including cutting fluids. Sensitization seems very rare.
Biochem/physiol Actions
Butylated hydroxytoluene is a phenolic antioxidant. It has been shown to inhibit lipid peroxidation. It causes lung injury and promotes tumors in mice, but this may be due to a metabolite of Butylated Hydroxytoluene, 6-tert-butyl-2-[2′-(2′-hydroxymethyl)-propyl]-4-methylphenol. Metabolites of Butylated Hydroxytoluene have also been reported to induce DNA strand breaks and internucleosomal DNA fragmentation (a characteristic of apoptosis) in cultured cells. In rats, a single intraperitoneal injection of Butylated Hydroxytoluene (60 mg/kg body mass) results in a significant increase in nuclear DNA methyl transferase activity in the liver, kidneys, heart, spleen, brain and lungs. Incubation of alveolar macrophages with Butylated Hydroxytoluene significantly reduced the level of TNF-α which may explain the mechanism by which this antioxidant reduces inflammation. Preincubation of aspirin-treated platelets with Butylated Hydroxytoluene inhibits the secretion, aggregation, and protein phosphorylation induced by protein kinase C activators. Butylated Hydroxytoluene was also found to inhibit the initiation of hepatocarcinogenesis by aflatoxin B1.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.
Butylated Hydroxytoluene Application
Butylated Hydroxytoluene (BHT) as a general antioxidant is used widely in polymer materials, petroleum products and food processing industries. It is commonly used rubber antioxidant, heat, oxygen aging have some protective effect, but also can inhibit copper harm.
Carcinogenicity
The IARC has determined that there is limited evidence for the carcinogenicity of Butylated Hydroxytoluene in experimental animals.Butylated Hydroxytoluene has given primarily negative results in a large number of in vivo and in vitro genotoxic assays.No significant reproductive effects were observed in three-generation toxicity studies in mice administered up to 0.4% in the diet.6 The 2003 ACGIH threshold limit valuetime- weighted average (TLV-TWA) for 2,6-ditert- butyl-p-cresol is 2mg/m3.
Environmental Fate
The metabolites of Butylated Hydroxytoluene can bind to cellular macromolecules, such as proteins and DNA, and cause toxicity.
Potential Exposure
DBPC is used as a food and feed additive, flavor, and packaging material; as an antioxidant in human foods and animal feeds. It is also used as an antioxidant to sta- bilize petroleum fuels, rubber and vinyl plastics.
Safety Profile
Poison by intraperitoneal andintravenous routes. Moderately toxic by ingestion. Anexperimental teratogen. Other experimental reproductiveeffects. A human skin irritant. A skin and eye irritant.Questionable carcinogen with experimental carcinogenicand.
Safety
Butylated hydroxytoluene is readily absorbed from the gastrointestinal tract and is metabolized and excreted in the urine mainly as glucuronide conjugates of oxidation products. Although there have been some isolated reports of adverse skin reactions, butylated hydroxytoluene is generally regarded as nonirritant and nonsensitizing at the levels employed as an antioxidant.
The WHO has set a temporary estimated acceptable daily intake for butylated hydroxytoluene at up to 125 μg/kg body-weight.
Ingestion of 4 g of butylated hydroxytoluene, although causing severe nausea and vomiting, has been reported to be nonfatal.
LD50 (guinea pig, oral): 10.7 g/kg
LD50 (mouse, IP): 0.14 g/kg
LD50 (mouse, IV): 0.18 g/kg
LD50 (mouse, oral): 0.65 g/kg
LD50 (rat, oral): 0.89 g/kg
Storage
Exposure to light, moisture, and heat causes discoloration and a loss of activity. Butylated hydroxytoluene should be stored in a wellclosed container, protected from light, in a cool, dry place.
Shipping
UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.
Purification Methods
Dissolve Butylated Hydroxytoluene in n-hexane at room temperature, then cool with rapid stirring, to -60o. The precipitate is separated, redissolved in hexane, and the process is repeated until the mother liquor is no longer coloured. The final product is stored under N2 at 0o [Blanchard J Am Chem Soc 82 2014 1960]. It has also been recrystallised from EtOH, MeOH, *benzene, n-hexane, methylcyclohexane or pet ether (b 60-80o), and is dried in a vacuum. [Beilstein 6 IV 3511.]
Toxicity evaluation
Butylated Hydroxytoluene is a white crystalline solid. It is insoluble in water and alkalies; but soluble in most common organic solvents such as alcohol and ether. Its melting point is 70°C, boiling point is 265°C, flash point is 127°C, and specific gravity is 1.048 at 20°C.
Incompatibilities
Butylated hydroxytoluene is phenolic and undergoes reactions characteristic of phenols. It is incompatible with strong oxidizing agents such as peroxides and permanganates. Contact with oxidizing agents may cause spontaneous combustion. Iron salts cause discoloration with loss of activity. Heating with catalytic amounts of acids causes rapid decomposition with the release of the flammable gas isobutene.
Regulatory Status
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM and IV injections, nasal sprays, oral capsules and tablets, rectal, topical, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Properties of Butylated hydroxytoluene (BHT)
| Melting point: | 69-73 °C(lit.) |
| Boiling point: | 265 °C(lit.) |
| Density | 1.048 |
| vapor density | 7.6 (vs air) |
| vapor pressure | <0.01 mm Hg ( 20 °C) |
| refractive index | 1.4859 |
| FEMA | 2184 | BUTYLATED HYDROXYTOLUENE |
| Flash point: | 127 °C |
| storage temp. | 2-8°C |
| solubility | methanol: 0.1 g/mL, clear, colorless |
| form | Crystals |
| pka | pKa 14(H2O
t = 25
c = 0.002 to 0.01) (Uncertain) |
| color | white |
| Odor | faint characteristic odor |
| Water Solubility | insoluble |
| Merck | 14,1548 |
| BRN | 1911640 |
| Exposure limits | ACGIH: TWA 2 mg/m3 NIOSH: TWA 10 mg/m3 |
| Stability: | Stable, but light-sensitive. Incompatible with acid chlorides, acid anhydrides, brass, copper, copper alloys, steel, bases, oxidizing agents. Combustible. |
| CAS DataBase Reference | 128-37-0(CAS DataBase Reference) |
| IARC | 3 (Vol. 40, Sup 7) 1987 |
| NIST Chemistry Reference | Butylated hydroxytoluene(128-37-0) |
| EPA Substance Registry System | 2,6-Di-tert-butyl-p-cresol (128-37-0) |
Safety information for Butylated hydroxytoluene (BHT)
| Signal word | Warning |
| Pictogram(s) |
 Environment GHS09 |
| GHS Hazard Statements |
H410:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P273:Avoid release to the environment. P391:Collect spillage. Hazardous to the aquatic environment P501:Dispose of contents/container to..… |
Computed Descriptors for Butylated hydroxytoluene (BHT)
| InChIKey | NLZUEZXRPGMBCV-UHFFFAOYSA-N |
Butylated hydroxytoluene (BHT) manufacturer
Kavya Pharma
Arcube Science
DL Intrachem
Atishay Specialities
R J Organics
Jaiswal S Cyber Shop
Sparkle Chem
New Products
AMino-(3-Methylphenyl)-acetic acid 3-AMINO-3-(2-CHLORO-PHENYL)-PROPIONIC ACID 4-Fluorophenylglycine 1-Boc-3-(cyanomethyl)azetidine 1-N-Boc-3-(aminoethyl)azetidine Dimedone 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 2-Bromo-5-Chloropyridine 2-Picolinic acid N-oxide 4-Butyl Resorcinol 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 5-Bromo-2-Hydroxy-3-Nitro Pyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside 4-Pentyn-1-ol 2-Bromo-6-fluoroaniline 5-fluoro-1,3-benzodioxole 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 2-(Chloromethyl) quinazolin-4(3H)-one 4-Chloro-2-methyl quinoline 2-Amino-4-phenyl-thiazoleRelated products of tetrahydrofuran

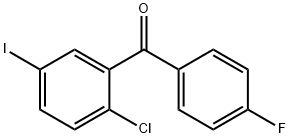

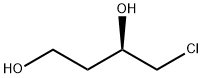
![(3S,3'S)-3,3'-((((4,4'-dichloro-[1,1'-biphenyl]-3,3'-diyl)bis (methylene))bis(4,1-phenylene))bis(oxy))bis(tetrahydrofuran)](https://img.chemicalbook.in/CAS/20200119/GIF/CB14713877.gif)
You may like
-
 BUTYLATED HYDROXY TOLUENE 99%View Details
BUTYLATED HYDROXY TOLUENE 99%View Details -
 2,6-Di-tert-butyl-4-methylphenol 98%View Details
2,6-Di-tert-butyl-4-methylphenol 98%View Details -
 Butylated Hydroxytoluene (BHT) CASView Details
Butylated Hydroxytoluene (BHT) CASView Details -
 Butylated Hydroxytoluene BHT CASView Details
Butylated Hydroxytoluene BHT CASView Details -
 Butylated Hydroxy Toluene CASView Details
Butylated Hydroxy Toluene CASView Details -
 Butylated Hydroxy Toluene (SQ) CAS 128-37-0View Details
Butylated Hydroxy Toluene (SQ) CAS 128-37-0View Details
128-37-0 -
 Butylated Hydroxy Toluene BHT CASView Details
Butylated Hydroxy Toluene BHT CASView Details -
 Butylated Hydroxytoluene BHT CASView Details
Butylated Hydroxytoluene BHT CASView Details
