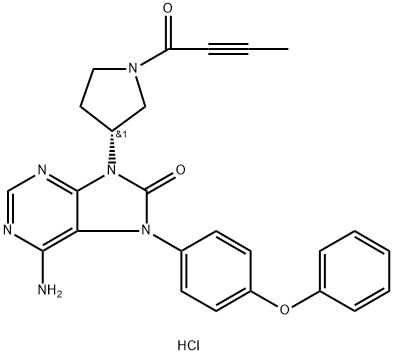
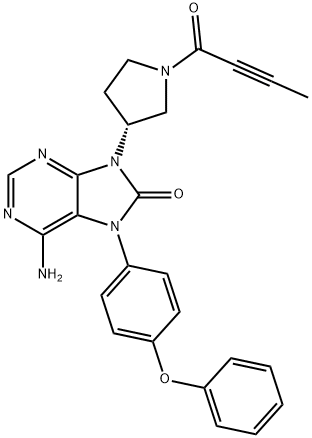
Tirabrutinib
- CAS NO.:1351636-18-4
- Empirical Formula: C25H22N6O3
- Molecular Weight: 454.48
- MDL number: MFCD28386296
- Update Date: 2026-04-17 10:13:23
What is Tirabrutinib?
Description
Btk Kinase inhibitor is highly potent and selective orally administered, small molecule, and also named tirabrutinib, Brutons tyrosine kinase (BTK) inhibitor being developed by Ono pharmaceutical and its licensee Gilead Sciences for the treatment of autoimmune disorders and haematological malignancies.
brand name
VelexbruTM
Biological Functions
In March 2020, tirabrutinib was approved in Japan to treat primary central nervous system lymphoma (PCNSL). Tirabrutinib shows excellent selectivity against all kinases with cysteines in the ATP-binding site corresponding to Cys481 in BTK with the exception of BMX, TXK, and TEC? However, it displays weaker BTK inhibition than ibrutinib (IC50 = 6.8 nM vs. 0.47 nM).
General Description
Class: non-receptor tyrosine kinase
Treatment: PCNSL
Elimination half-life = 6.5–8 h
Protein binding = 91%
Mechanism of action
The selective inhibition of cell growth by Btk Kinase inhibitor was due to blocking of BTK-mediated signaling through AKT and cellular protein kinase D. It can inhibit autophosphorylation of the BTK at the Tyr223 position through the ERK, AKT and PKD signaling pathways.
Side Effects
Adverse effects of the Btk Kinase inhibitor that occurred in some patients were rash, vomiting, neutropenia, arthralgia, and malaise, and drug-related Grade 3–4 Adverse effects were neutropenia, leukopenia, anemia, hypophosphatemia, PT-INR increased, pneumonitis, and acute myeloid leukemia. in prior Japanese studies, rash, hematologic adverse effects, erythema multiforme, and constipation were frequent adverse effects in a Phase I/II study of some patients with PCNSL, and rash, hematologic adverse effects, and stomatitis were the most common adverse effects in a Phase II study of some patients with WM.
Properties of Tirabrutinib
| Boiling point: | 672.0±65.0 °C(Predicted) |
| Density | 1.412±0.06 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMSO:65.0(Max Conc. mg/mL);143.02(Max Conc. mM) DMSO:PBS (pH 7.2) (1:2):0.3(Max Conc. mg/mL);0.66(Max Conc. mM) DMF:30.0(Max Conc. mg/mL);66.01(Max Conc. mM) Ethanol:1.0(Max Conc. mg/mL);2.2(Max Conc. mM) |
| form | A crystalline solid |
| pka | 3.16±0.20(Predicted) |
| color | White to yellow |
Safety information for Tirabrutinib
Computed Descriptors for Tirabrutinib
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran
You may like
-
 88660-53-1 >99%View Details
88660-53-1 >99%View Details
88660-53-1 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4