Benzyl vinylcarbamate
- CAS NO.:84713-20-2
- Empirical Formula: C10H11NO2
- Molecular Weight: 177.2
- MDL number: MFCD04114067
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-07-24 18:13:54
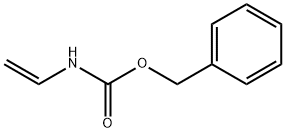
What is Benzyl vinylcarbamate?
The Uses of Benzyl vinylcarbamate
Benzyl Vinylcarbamate is used as a reagent in the synthesis of CF3- or -?CF2-?substituted tetrahydroquinolines.
Preparation
A 1-L flask was equipped with a variable speed pump, a mechanical stirrer, a temperature controller, a 4" (10 cm) column packed with ceramic saddles, a distillation head, a spiral condenser (cooled with water at 10–15 ℃), and a receiver. The flask was charged with toluene (150–200 mL) and phenothiazine (0.5 g) and the solution was heated to 105–110 ℃. The receiver was charged with benzyl alcohol (86 g, 0.8 mol), phenothiazine (0.05 g), and triethylamine (0.1–0.3 g). This mixture was cooled in ice and stirred. A solution of acryloyl azide (1 mol), prepared as described above, was pumped into the distillation flask over a period of 4–5 h, maintaining the pot temperature at 105–110 ℃ with a heating mantle. The vapor temperature varied, depending on the rate of addition of the azide, but was in the range 80–100 ℃. The distillate was passed directly into the benzyl alcohol mixture. After the addition of acryloyl azide, the distillation continued, generating a further 10–20 mL of toluene. The receiver was then removed from the distillation set-up, and its contents were stirred at 0–5 ℃ for 1– 2 h. The product mixture was then allowed to gradually warm to room temperature and was stirred until HPLC analysis indicated complete reaction. The mixture was then concentrated in vacuo to a weight of 200–250 g. The residue was treated with heptane (300–350 mL) and cooled to 15 ℃ with stirring. A few seed crystals of benzyl N-vinyl carbamate 810 were added, and the mixture was stirred for 2–3 h. The product was collected by filtration, washed with heptane, and dried in vacuo. Yield 115–128 g (65–72%); mp 41–44 ℃.
What are the applications of Application
Benzyl-N-vinyl carbamate (Z-vinylamine; Benzyl vinylcarbamate) is a valuable synthetic intermediate. This compound undergoes alkylation readily on the carbon R to the nitrogen, a property that has been used in the synthesis of a-lactam antibiotics. Z-vinylamine can be readily polymerized into polyvinyl amine derivatives[1].
Synthesis Reference(s)
The Journal of Organic Chemistry, 54, p. 4649, 1989 DOI: 10.1021/jo00280a036
References
[1] Govindan, Cheruthur K. “An Improved Process for the Preparation of Benzyl-N-vinyl Carbamate1.” Organic Process Research Development 6 1 (2001): 74–77.
Properties of Benzyl vinylcarbamate
| Melting point: | 43-44°C |
| Boiling point: | 272℃ |
| Density | 1.089 |
| Flash point: | 118℃ |
| storage temp. | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| solubility | Chloroform (Slightly), Ethyl Acetate (Slightly) |
| form | Solid |
| pka | 11.04±0.46(Predicted) |
| color | White to Off-White Low-Melting |
Safety information for Benzyl vinylcarbamate
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Benzyl vinylcarbamate
New Products
Dexamethasone Sodium Phosphate p-Anisaldehyde m-Tolualdehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid Raloxifene EP Impurity D N-Nitroso Nortriptyline Nitroso Irbesartan 2-(p-Tolyl)acetaldehyde Sulfonic acid Impurity 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 5-Chloro-2-Methoxy-4-Picoline 2-Amino-6-nitrobenzoic Acid (6-Nitroanthranilic acid) Cis-3,5 Diacetoxy-1-cyclopentene 3-Bromo-2-Methoxy-4-Methylpyridine 2-Amino-5-Chloro-3-Nitro-6-picoline 3-Methoxy-5-methylpyrazin-2-amine Pentachlorobenzonitrile Budesonide Nicotinic acid RAPAMYCIN Alpha CyclodextrinRelated products of tetrahydrofuran
![3-CHLOROBENZYL N-[2-(2-FURYL)VINYL]CARBAMATE](https://img.chemicalbook.in/StructureFile/ChemBookStructure3/GIF/CB9724909.gif)


![BENZYL N-[2-(2-FURYL)VINYL]CARBAMATE](https://img.chemicalbook.in/StructureFile/ChemBookStructure3/GIF/CB0315555.gif)
![4-CHLOROBENZYL N-[2-(2-FURYL)VINYL]CARBAMATE](https://img.chemicalbook.in/StructureFile/ChemBookStructure3/GIF/CB6454652.gif)


You may like
-
 Benzyl vinylcarbamate 95% CAS 84713-20-2View Details
Benzyl vinylcarbamate 95% CAS 84713-20-2View Details
84713-20-2 -
 12029-98-0 >98%View Details
12029-98-0 >98%View Details
12029-98-0 -
 1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 Dexamethasone Acetate >98%View Details
1177-87-3 -
 Hydrocortisone 50-23-7 >98%View Details
Hydrocortisone 50-23-7 >98%View Details
50-23-7 -
 106-43-4 Parachlorotoluene >99%View Details
106-43-4 Parachlorotoluene >99%View Details
106-43-4 -
 Pyrrolidin >98%View Details
Pyrrolidin >98%View Details
123-75-1 -
 cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
cis-p-Menth-2,8-diene-1-ol 7212-40-0 >98%View Details
7212-40-0 -
 Para Chloro toluene (PCT) 106-43-4 >99%View Details
Para Chloro toluene (PCT) 106-43-4 >99%View Details
106-43-4
