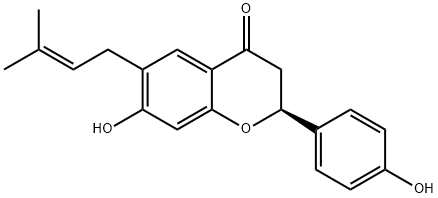
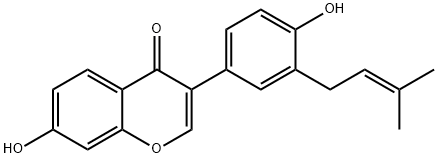
BAVACHIN
Synonym(s):(2S)-2,3-Dihydro-7-hydroxy-2-(4-hydroxyphenyl)-6-(3-methyl-2-butenyl)-4H-1-benzopyran-4-one;4′,7-Dihydroxy 6-(3-methyl 2-butenyl)flavanone;Corylifolin
- CAS NO.:19879-32-4
- Empirical Formula: C20H20O4
- Molecular Weight: 324.37
- MDL number: MFCD11617359
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 12:01:10
What is BAVACHIN?
Description
Bavachin is a flavonoid first isolated from seeds of P. corylifolia. It is a phytoestrogen that activates the estrogen receptors ERα and ERβ (EC50s = 320 and 680 nM, respectively). Through this action, bavachin stimulates osteoblast proliferation and differentiation and prevents bone loss following ovariectomy in rats. Bavachin less potently inhibits acyl-coenzyme A:cholesterol acyltransferase (IC50 = 86 μM).
The Uses of BAVACHIN
Bavachin has therapeutic potential for type 2 diabetes by activating insulin signaling pathways. Also, it has anabolic and potent anticatabolic biological effects on chondrocytes. Strong inhibitor of human UDP-glucuronosyltransferase (UGT1A1)
What are the applications of Application
Bavachin is a weak antioxidant that stimulates bone formation
References
[1] choi j h, rho m c, lee s w, et al. bavachin and isobavachalcone, acyl-coenzyme a: cholesterol acyltransferase inhibitors from psoralea corylifolia[j]. archives of pharmacal research, 2008, 31(11): 1419-1423.
[2] park j, kim d h, ahn h n, et al. activation of estrogen receptor by bavachin from psoralea corylifolia[j]. biomolecules & therapeutics, 2012, 20(2): 183-188.
[3] lee h, li h, noh m, et al. bavachin from psoralea corylifolia improves insulin-dependent glucose uptake through insulin signaling and ampk activation in 3t3-l1 adipocytes[j]. international journal of molecular sciences, 2016, 17(4): 527.
Properties of BAVACHIN
| Boiling point: | 558.3±50.0 °C(Predicted) |
| Density | 1.244 |
| storage temp. | Inert atmosphere,Store in freezer, under -20°C |
| solubility | ≤20mg/ml in ethanol;30mg/ml in DMSO;50mg/ml in dimethyl formamide |
| form | crystalline solid |
| pka | 8.27±0.40(Predicted) |
| color | White |
| CAS DataBase Reference | 19879-32-4 |
Safety information for BAVACHIN
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. |
Computed Descriptors for BAVACHIN
New Products
2-Ethoxyphenol Methyl 2-methoxy-5-sulfamoylbenzoate Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 3,6-Dichloropyridazine 3,6-Dichloro-4-Isopropylpyridazine 2-[(2-Ethoxyphenoxy)methyl]oxirane m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 6-Methyl-2-pyridinemethanol 2-Pyridinecarboxaldehdye tert-Butyl ((1R,2S,5S)-2-amino-5-(dimethylcarbamoyl)cyclohexyl)carbamate 1-Indanone 2-Chloromethyl-6-methyl-pyridine 4-Morpholinoaniline Abiraterone Ethyl Ether 1-nitro-3,5-dimethyl adamantine (NMEM) Vardenafil Bis-sulphonamide(Dimer) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-oneRelated products of tetrahydrofuran
You may like
-
 Bavachin CAS 19879-32-4View Details
Bavachin CAS 19879-32-4View Details
19879-32-4 -
![3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 99%+View Details
2304754-51-4 -
 1126-74-5 99%+View Details
1126-74-5 99%+View Details
1126-74-5 -
 2-Fluoro-6-iodobenzoic acid 99%+View Details
2-Fluoro-6-iodobenzoic acid 99%+View Details
111771-08-5 -
 120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride 99%+View Details
120013-39-0 -
 4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
4-FORMYL-N-CBZ-PIPERIDINE 99%+View Details
138163-08-3 -
![ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+](https://img.chemicalbook.in//Content/image/CP5.jpg) ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 313544-31-9 99%+View Details
313544-31-9 -
 1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
1-Boc-4-cyanopiperidine 91419-52-2 99%+View Details
91419-52-2
