Apcin
Synonym(s):2-(2-Methyl-5-nitroimidazol-1-yl)ethyl N-[2,2,2-trichloro-1-(pyrimidin-2-ylamino)ethyl]carbamate;3-(2-Methyl-5-nitro-imidazol-1-yl)-N-(2,2,2-trichloro-1-phenylamino-ethyl)-propionamide
- CAS NO.:300815-04-7
- Empirical Formula: C13H14Cl3N7O4
- Molecular Weight: 438.65
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-03-13 23:13:44
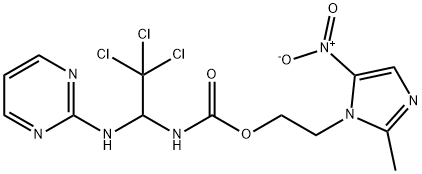
What is Apcin?
The Uses of Apcin
Apcin is a novel inhibitor of the anaphase-promoting complex/cyclosome (APC/C), blocking mitotic exit and being synergistically amplified by co-addition of Ts-Arg-OMe.
Biochem/physiol Actions
Apcin is an inhibitor of the anaphase-promoting complex/cyclosome (APC/C), a large multimeric complex that functions as a ubiquitin ligase, initiating the metaphase–anaphase transition and regulating ordered transitions through the cell cycle by controlling the ubiquitin-mediated proteolysis of cell cycle proteins. Apcin prevents substrate recognition and inhibits APC/C-dependent ubiquitylation by binding to the D-box binding site of Cdc20, a coactivator required for full activation of APC/C and substrate recognition. The effectiveness of apcin in blocking mitotic exit was synergistically amplified by co-addition of TAME (tosyl-L-arginine methyl ester) or its cell permeable prodrug proTAME, which block Cdc20 binding to the APC/C at a different site than apcin.
Enzyme inhibitor
This Cdc20 inhibitor (FW = 438.65 g/mol; CAS 300815-04-7; Solubility: 100 mM in DMSO), also named 3-(2-methyl-5-nitroimidazol-1-yl)-N- (2,2,2-trichloro-1-phenylaminoethyl)propionamide, targets substrate interaction of Cell division control protein 20, blocking substrate-mediated Cdc20 loading onto the Anaphase-Promoting Complex. APC is a 13- subunit ubiquitin ligase that initiates the metaphase-anaphase transition and mitotic exit by targeting proteins such as securin and cyclin B1 for ubiquitin-dependent destruction by the proteasome. APC regulates cell cycle progression by forming APC·Cdc20 and APC·Cdh1, two closely related, but functionally distinct, E3 ubiquitin ligase sub-complexes. Cdh1 and Cdc20 have opposing functions in tumorigenesis, with the former largely functioning as a tumor suppressor, and the latter exhibiting an oncogenic function. Because blocking mitotic exit is an effective approach for inducing tumour cell death, the APC/C represents a potential novel target for cancer therapy. APC/C activation during mitosis requires Cdc20binding, forming a co-receptor with the APC/C to recognize substrates containing a destruction box (D-box). APC/C-dependent proteolysis and mitotic exit can be synergistically inhibited by simultaneously disrupting two protein-protein interactions within the APC/C-Cdc20-substrate ternary complex. APC inhibitor, or Apcin, binds to Cdc20, competitively inhibiting ubiquitylation of D-box-containing substrates. Analysis of the crystal structure of the apcin-Cdc20 complex suggests that apcin occupies the D-box-binding pocket on the side face of the WD40-domain. The ability of Apcin to block mitotic exit is enhanced by co-addition of tosyl-l-arginine methyl ester (See TAME), a small molecule that blocks the APC/C-Cdc20 interaction. Apcin also inhibits APC-dependent ubiquitylation and prolongs mitotic duration in RPE1 cells, when administered in combination with proTAME, a prodrug of TAME in vitro (See pro-TAME).
Storage
Store at -20°C
Properties of Apcin
| Density | 1.68±0.1 g/cm3(Predicted) |
| storage temp. | room temp |
| solubility | DMF: 25 mg/ml; DMSO: 25 mg/ml; DMSO:PBS (pH 7.2) (1:1): 0.5 mg/ml |
| form | powder |
| pka | 9.67±0.46(Predicted) |
| color | white to beige |
Safety information for Apcin
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Apcin
New Products
Ivermectin Ep Impurity- k Cyclopropane-1,1-dicarboxylic acid [1]Benzothieno[2,3-c]pyridine, 1,7-dichloro-8-methyl- (S)-3-Fluoro-pyrrolidine hydrochloride Cyclobutylamine 1-Bromo-2-Methoxy-3-Nitrobenzene 3-Oxocyclobutane-1-carboxylicacid (4-Bromophenyl)(3,4-dimethoxyphenyl)methanone N-Nitroso hydroxy Cetrizine EP Impurity-A Noradrenaline EP Impurity D/Noradrenaline Methyl Ether Cetirizine EP Impurity A/Cetirizine CBHP Impurity Lantanoprost rc B Clidinium Bromide Impurity 2-chloro-7-cyclopentyl-N,N-dimethylpyrrolo [2,3-d]pyrimidine-6-carbox amide 4-amino-2-fluoro-N- methylbenzamide N-(2-chloropyrimidin-4-yl)-N,2,3- trimethyl-2H- indazol 6-amine 5-Chloro-3- Methyl -1,3-2-benzoxazol-2(3H)-one 4-Bromo-2-fluoro-N-methylbenzamide N-Boc-1,4-butanediamine 4-Dimethylamino pyridine 2-Chlorotrityl Chloride 4-Bromoaniline 5-Bromo-2-chlorobenzoic acid 1,2,3,9-Tetrahydro-4(H)-carbazol-4-oneYou may like
-
 Apcin >98% (HPLC) CAS 300815-04-7View Details
Apcin >98% (HPLC) CAS 300815-04-7View Details
300815-04-7 -
 Apcin CAS 300815-04-7View Details
Apcin CAS 300815-04-7View Details
300815-04-7 -
![3-(4-bromo-3-methyl-2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 98+](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-(4-bromo-3-methyl-2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 98+View Details
3-(4-bromo-3-methyl-2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2304754-51-4 98+View Details
2304754-51-4 -
 KT-474 98+View Details
KT-474 98+View Details
2432994-31-3 -
![2,6-Piperidinedione, 3-hydroxy-1-[(4-methoxyphenyl)methyl]- 98+](https://img.chemicalbook.in//Content/image/CP5.jpg) 2,6-Piperidinedione, 3-hydroxy-1-[(4-methoxyphenyl)methyl]- 98+View Details
2,6-Piperidinedione, 3-hydroxy-1-[(4-methoxyphenyl)methyl]- 98+View Details
2357109-89-6 -
 2,3-Difluoro-6-methoxybenzyl Chloride 98+View Details
2,3-Difluoro-6-methoxybenzyl Chloride 98+View Details
1073435-67-2 -
 2-(6-(benzyloxy)-3,4-dihydronaphthalen-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane 2477812-43-2 98+View Details
2-(6-(benzyloxy)-3,4-dihydronaphthalen-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane 2477812-43-2 98+View Details
2477812-43-2 -
 101894-09-1 4-Hydroxy-6-methyl-1-(4-methylphenyl)-2-naphthalenecarboxylic acid 98+View Details
101894-09-1 4-Hydroxy-6-methyl-1-(4-methylphenyl)-2-naphthalenecarboxylic acid 98+View Details
101894-09-1
