Ambazone
- CAS NO.:539-21-9
- Empirical Formula: C8H11N7S
- Molecular Weight: 237.28
- EINECS: 208-713-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:08:57
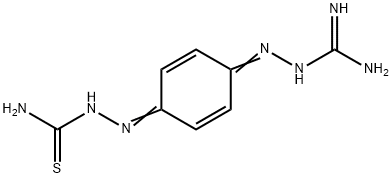
What is Ambazone?
Originator
Ambazone,Generic
The Uses of Ambazone
Ambazone is a benzoquidine diimine derivative useful as animal growth substance.
What are the applications of Application
Ambazone is Ambazone is a benzoquidine diimine derivative which has shown to have antiseptic and anti-tumor properties.
Definition
ChEBI: Ambazone is a hydrazone.
Manufacturing Process
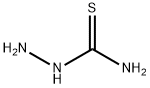
To a solution of 22.7 g of quinone monoguanylhydrazone nitrate in 250 ml water is added dropwise hot aqueous solution of 9.1 g thiosemicarbazide, then is added slowly a solution of 5 ml concentrated nitric acid in 10 ml of water. The mixture is stirred at 60°C for 1 hour. The product is dissolved in 1-1.2 L of water at 100°C. The solution is filtered and added to an aqueous ammonium solution. Blue residue of p-benzoquinone amidinohydrazone thiosemicarbazone is filtered and dried, melting point 188°C (decomp.).
Therapeutic Function
Antiseptic
Properties of Ambazone
| Melting point: | 195°C (rough estimate) |
| Boiling point: | 160°C (rough estimate) |
| Density | 1.2732 (rough estimate) |
| refractive index | 1.5500 (estimate) |
| storage temp. | Hygroscopic, Refrigerator, under inert atmosphere |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| form | Solid |
| color | Red to Very Dark Brown |
| Stability: | Very Hygroscopic |
| EPA Substance Registry System | Hydrazinecarbothioamide, 2-[4-[(aminoiminomethyl)hydrazono]-2,5-cyclohexadien-1-ylidene]- (539-21-9) |
Safety information for Ambazone
Computed Descriptors for Ambazone
New Products
Methyl N-((2'-cyano-[1,1'-biphenyl]-4-yl)methyl)-N-nitroso-L-valinate Levofloxacin - Impurity B/ Levofloxacin Related Compound A Ivabradine R isomer Escitalopram - Impurity K Escitalopram Cyno dilol Impurity Paroxetine EP impurity I/Paroxetine Anhydrous EP Impurity I 2-Amino-5-Bromo-6-Chloropyridine 3-Amino-5-bromopicolinic acid 1-(7,8-Dinitro-4,5-dihydro-1H-1,5-methanobenzo[d]azepin-3(2H)-yl)-2,2,2-trifluoroethanone 2-Amino-3-Bromo-5-Cyanopyridine 2-Amino-3-Chloropyridine 2,5-Dibromo-3-MethylpyridineRelated products of tetrahydrofuran

You may like
-
 Benzoquinone Guanylhydrazone Thiosemicarbazone, 99.50%View Details
Benzoquinone Guanylhydrazone Thiosemicarbazone, 99.50%View Details
106-51-4 -
 17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 Diphenhydramine EP impurity-A /Diphenhydramine Related Compound A NLT 95%View Details
17471-10-2 -
 Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
Dorzolamide Related Compound B/Dorzolamide EP Impurity B NLT 95%View Details
120279-90-5 -
 76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 Famotidine - Impurity D NLT 95%View Details
76824-16-3 -
 118-23-0 NLT 95%View Details
118-23-0 NLT 95%View Details
118-23-0 -
 Miconazole EP Impurity F/ Miconazole USP Related Compound F 47447-55-2 NLT 95%View Details
Miconazole EP Impurity F/ Miconazole USP Related Compound F 47447-55-2 NLT 95%View Details
47447-55-2 -
 2407630-84-4 NLT 95%View Details
2407630-84-4 NLT 95%View Details
2407630-84-4 -
 573704-41-3 NLT 95%View Details
573704-41-3 NLT 95%View Details
573704-41-3
