Aflibercept
- CAS NO.:845771-78-0
- Molecular Weight: 0
- Update Date: 2026-03-18 11:52:23
What is Aflibercept?
Mechanism of action
Aflibercept is a 115 kDa fusion protein. It consists of an IgG backbone fused to extracellular VEGF receptor sequences of the human VEGFR1 and VEGFR2. As a soluble decoy receptor, it binds VEGF-A with a greater affinity than its natural receptors. In an experimental model, aflibercept’s equilibrium disassociation constant (Kd, inversely related to binding affinity) for VEGF-A165 was 0.49 pM, compared with 9.33 pM and 88.8 pM for experimental native VEGFR1 and VEGFR2, respectively. Aflibercept’s high affinity for VEGF prevents the subsequent binding and activation of native VEGF receptors. Reduced VEGF activity leads to decreased angiogenesis and vascular permeability. Inhibition of PIGF and VEGF-B may also aid the treatment of angiogenic conditions. PIGF has been associated with angiogenesis and can be elevated in multiple conditions, such as wet AMD. VEGF-B overexpression has been connected with the breakdown of the blood-retinal barrier and retinal angiogenesis. Thus, inhibition of VEGF-A, VEGF-B, and PIGF may all contribute to the efficacy of aflibercept. Aflibercept has a unique binding action and binds to both sides of the VEGF dimer, forming an inert 1:1 complex called a VEGF trap. Additionally, aflibercept is the only drug in its class that binds to PIGF-2.
Clinical Use
Antineoplastic agent:
Treatment of metastatic colorectal cancer
Clinical Use
Ophthalmology
Intravitreal aflibercept injection was approved by the FDA in 2011 for the treatment of neovascular (wet) age-related macular degeneration (AMD) following two large clinical trials. Since then, it has also been approved for macular edema following retinal vein occlusion (RVO), diabetic macular edema (DME), diabetic retinopathy (DR) in patients with DME, and, most recently, for retinopathy of prematurity (ROP).
Oncology
Ziv-aflibercept, in combination with 5-fluorouracil, leucovorin, and irinotecan-(FOLFIRI), is indicated for patients with metastatic colorectal cancer (mCRC) that is resistant to or has progressed following an oxaliplatin?containing regimen. Ziv-aflibercept contains the same protein (active drug) as aflibercept but is specifically formulated for injection as an intravenous infusion. Ziv-aflibercept is not intended for ophthalmic use, as the osmolarity of the ziv-aflibercept preparation is significantly higher than that of intravitreal aflibercept injection. However, intravitreal ziv-aflibercept has been used with success for multiple ocular conditions with an acceptable safety profile.
Metabolism
No metabolism studies have been conducted with aflibercept since it is a protein. Aflibercept is expected to degrade to small peptides and individual amino acids. Free aflibercept is mainly cleared by binding to endogenous VEGF to form a stable, inactive complex. As with other large proteins, both free and bound aflibercept, are expected to be cleared, more slowly, by other biological mechanisms, such as proteolytic catabolism.
Properties of Aflibercept
| form | Liquid |
| color | Colorless to light yellow |
Safety information for Aflibercept
Computed Descriptors for Aflibercept
New Products
3-azanyl-3-phenyl-propanoic acid 3-AMINO-3-(2-METHYLPHENYL)PROPANOIC ACID 3-AMINO-3-M-TOLYL-PROPIONIC ACID Nitroso Irbesartan N-Nitroso Nortriptyline 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 4-dimethylaminopyridine 3-Bromo-2-Methyl-5-Nitropyridine 2-Chloro Isonicotinic acid methyl ester 2-(Ethoxymethylene)propanedinitrile 5-Bromo-3-Methyl-2-Pyridinecarboxylic acid 5-Chloro-pyridine-2-carboxylic acid amideRelated products of tetrahydrofuran
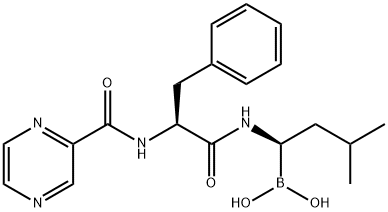



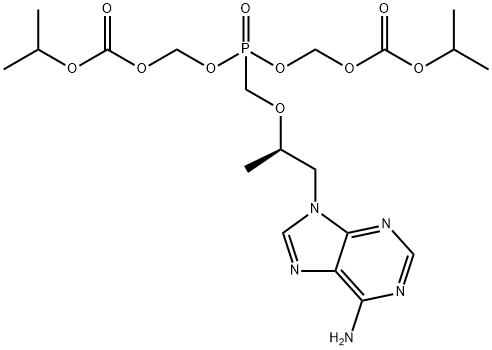
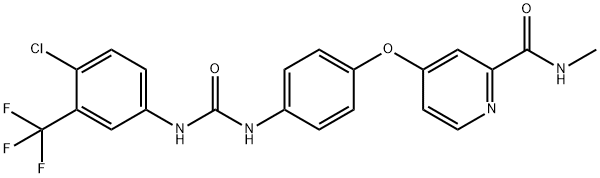
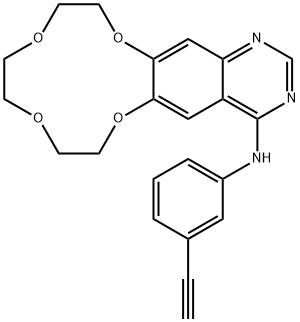
You may like
-
 3646-50-2 3-azanyl-3-phenyl-propanoic acid 98+View Details
3646-50-2 3-azanyl-3-phenyl-propanoic acid 98+View Details
3646-50-2 -
 68208-16-2 98+View Details
68208-16-2 98+View Details
68208-16-2 -
 3646-50-2 3-azanyl-3-phenyl-propanoic acid 98+View Details
3646-50-2 3-azanyl-3-phenyl-propanoic acid 98+View Details
3646-50-2 -
 68208-16-2 98+View Details
68208-16-2 98+View Details
68208-16-2 -
 3-AMINO-3-M-TOLYL-PROPIONIC ACID 98+View Details
3-AMINO-3-M-TOLYL-PROPIONIC ACID 98+View Details
68208-17-3 -
 3646-50-2 3-azanyl-3-phenyl-propanoic acid 98+View Details
3646-50-2 3-azanyl-3-phenyl-propanoic acid 98+View Details
3646-50-2 -
 68208-16-2 98+View Details
68208-16-2 98+View Details
68208-16-2 -
 3-AMINO-3-M-TOLYL-PROPIONIC ACID 98+View Details
3-AMINO-3-M-TOLYL-PROPIONIC ACID 98+View Details
68208-17-3
