Acodazole
- CAS NO.:55435-65-9
- Empirical Formula: C20H20ClN5O
- Molecular Weight: 381.8587
- MDL number: MFCD01670903
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-21 10:31:43
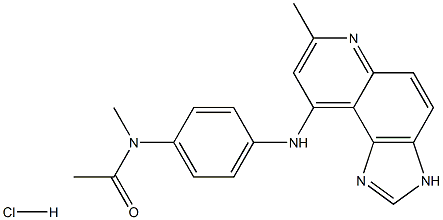
What is Acodazole?
Originator
Acodazole Hydrochloride ,ZYF Pharm
The Uses of Acodazole
Antineoplastic.
Manufacturing Process
An 82.0 g (0.5 mole) of 5-nitrobenzimidazole in 900 ml of ethanol was
reduced over 4.0 g of 5% Pd/C catalyst containing 50% H2O. After filtration of
the catalyst, 65.0 g (0.5 mole) of ethyl acetoacetate, 20.0 g of anhydrous
calcium sulfate, and 0.5 ml of HOAc was added. After filtration, the solution
was concentrated in vacuo till a solid remained. The product was filtered and
washed with fresh ethanol and air-dried. The yield of ethyl 3-(5-
benzimidazolylamino)crotonate was 84.0 g (69%), melting point 160°-162°C.
40.0 g of ethyl 3-(5-benzimidazolylamino)crotonate was added to 80 ml of
boiling Dowtherm? and the boiling was continued for 5 min. The product
separated upon cooling. The product was filtered, washed with Dowtherm?
and then acetone and air-dried. The yield of 7-methyl-9-imidazo[4,5-
f]quinolinol was 29.0 g (91%), melting point 345°-347°C.
Into a 22 L, 4-necked flask set in a tub and equipped with a stirrer, an air
condenser (drying tube), thermometer, and dropping funnel was placed POCl3
(4590 ml). The 7-methyl-9-imidazo[4,5-f]quinolinol (1062.0 g, 5.33 moles)
was added with no heating effect noted. Dimethylformamide (4690 ml) was
added dropwise over a 2.5 h period at a rate to control the temperature below
85°C. The resulting viscous solution was allowed to stand overnight at room
temperature and then added cautiously to ice to a total volume of ca. 50 L.
The resulting solution was then adjusted to a pH of 7 to 8 by the addition of
NaOH pellets (9771.0 g). More ice was added as needed to keep the
temperature below 45°C. The resulting precipitate was collected by filtration,
washed well by stirring in water (3x20 L) and dried at 60°C to yield 1107.0 g
(95.5%) of 9-chloro-7-methylimidazo[4,5-f]quinolone.
To 500 ml of acetic anhydride was added portionwise, 100.0 g (0.658) of Nmethyl-
p-nitroaniline. Following the addition, the solution was heated on a
steam bath for 2 h, then stirred overnight at room temperature. The white
precipitate of the N-methyl-4-nitroacetanilide was collected by filtration,
washed with ether and air-dried to give 53.0 g, melting point 153°-156°C.
The filtrate was concentrated in vacuum to give another 61.0 g, melting point
150°-154°C.
A mixture of 114.0 g (0.587 m) of N-methyl-4-nitroacetanilide and 800 ml of
ethanol was shaken with hydrogen over one teaspoon of Raney active nickel
catalyst in water. A pressure drop of 127 psi was recorded (calc. 118 psi). The
catalyst was removed by filtration and the ethanol filtrate refluxed overnight
with 127.0 g (0.587 m) of the 9-chloro-7-methylimidazo[4,5-f]quinolone. The
mixture was chilled, filtered, washed with ether and air-dried to give 75.5 g of
9-[p-(N-methylacetamido)anilino]-7-methyl-1-H-imidazo[4,5-f]quinoline hydrochloride sesquihydrate, melting point 315°-318°C (recrystallized from
4,000 ml of MeOH).
By treatment of 9-p-(N-methylacetamido)anilino-7-methyl-1H-imidazo[4,5-
f]quinolone hydrochloride sesquihydrate with NaOH may be produced the 9-p-
(N-methylacetamido)anilino-7-methyl-1H-imidazo[4,5-f]quinolone.
Therapeutic Function
Antineoplastic
Properties of Acodazole
| Melting point: | 315-317 °C |
Safety information for Acodazole
Computed Descriptors for Acodazole
New Products
Dexamethasone Sodium Phosphate 2-(2-Ethoxyphenoxy)ethyl bromide 8-Bromo-7-(2-butynyl)-3-methyl xanthine 5-Chlorothiophene-2-carboxylic acid m-Tolualdehyde p-Anisaldehyde 2-Bromobenzaldehyde 4-Fluorobenzaldehyde 1-Propyl-4-piperidone 3-Amino-3-(3-fluorophenyl)propanoic acid 2-Fluoro-6-iodobenzoic acid 3-(4-Bromo-3-methyl-2-oxo-2,3-dihydro-1h-benzo[d]imidazol-1-yl)piperidine-2,6-dione 2,3-Dihydro-5,6-dimethoxy-2-(4-piperidinylmethyl)-1H-inden-1-one hydrochloride ethyl 2-oxo-2,3,9,10-tetrahydro-1H-pyrido[3',4':4,5]pyrrolo[1,2,3-de]quinoxaline-8(7H)-carboxylate 1-Boc-4-cyanopiperidine 3-Pyridineacrylic acid Abiraterone Ethyl Ether Vardenafil Bis-sulphonamide(Dimer) 1-nitro-3,5-dimethyl adamantine (NMEM) 3-(2-aminoethyl) benzene sulfonamide Nabumetone Impurity 5 2,2-dibromo-1-cyclopropyl-2-(2-fluorophenyl)ethan-1-one 2-Amino-3,5-Dichloro-4-picoline 5-Amino-2-EthoxypyridineYou may like
-
 141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 3,6-Dichloropyridazine 98%View Details
141-30-0 -
 Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
Ethyl 2-chloro-2-(2-(4-methoxyphenyl)hydrazono)acetate 27143-07-3 98%View Details
27143-07-3 -
 94-71-3 98%View Details
94-71-3 98%View Details
94-71-3 -
 Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
Methyl 2-methoxy-5-sulfamoylbenzoate 33045-52-2 98%View Details
33045-52-2 -
 88965-00-8 98%View Details
88965-00-8 98%View Details
88965-00-8 -
 3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
3,6-Dichloro-4-Isopropylpyridazine 107228-51-3 98%View Details
107228-51-3 -
 4640-67-9 98%View Details
4640-67-9 98%View Details
4640-67-9 -
![2-[(2-Ethoxyphenoxy)methyl]oxirane 98%](https://img.chemicalbook.in//Content/image/CP5.jpg) 2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
2-[(2-Ethoxyphenoxy)methyl]oxirane 98%View Details
5296-35-5
