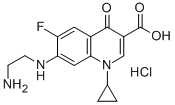
6-Hydroxy-6-defluoro Ciprofloxacin
- CAS NO.:226903-07-7
- Empirical Formula: C17H19N3O4
- Molecular Weight: 329.35
- MDL number: MFCD18379447
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-28 02:14:25
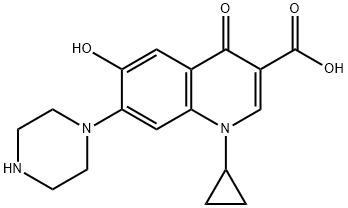
What is 6-Hydroxy-6-defluoro Ciprofloxacin?
Chemical properties
Off-White to Pale Beige Solid
The Uses of 6-Hydroxy-6-defluoro Ciprofloxacin
6-Hydroxy-6-defluoro Ciprofloxacin (Ciprofloxacin EP Impurity F) is a hydroxy analog of Ciprofloxacin (C482500).
Properties of 6-Hydroxy-6-defluoro Ciprofloxacin
| Melting point: | >258°C (dec.) |
| Boiling point: | 636.2±55.0 °C(Predicted) |
| Density | 1.490±0.06 g/cm3(Predicted) |
| storage temp. | -20°C Freezer |
| solubility | DMSO, Methanol |
| form | Solid |
| pka | 6.47±0.41(Predicted) |
| color | Off-White to Pale Beige |
| Stability: | Hygroscopic |
Safety information for 6-Hydroxy-6-defluoro Ciprofloxacin
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 6-Hydroxy-6-defluoro Ciprofloxacin
6-Hydroxy-6-defluoro Ciprofloxacin manufacturer
GLP Pharma Standards
3Y
Phone:+91-7893074638
Whatsapp: +91-7893074638
product: Ciprofloxacin Impurity-F 226903-07-7 NLT 95%
Clickchem Research LLP
3Y
Phone:+91-8140031133
Whatsapp: +91- 8140031133
product: CIPROFLOXACIN EP IMPURITY F 226903-07-7 90 % Above
New Products
Tridecanoic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 1-Fluorocyclopropanecarboxylic Acid 2-(tert-Butoxycarbonyl)-5,7-dichloro-1,2,3,4-tetrahydroisoquinoline-6-carboxylic acid (S)-3-Hydroxypyrrolidine hydrochloride Dimethyl (2,2-dihydroxy-3,3-difluoro-2-oxoheptyl)phosphonate Lead tetraacetate 7-iodopyrrolo[2,1-f][1,2,4]triazin-4-amine 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole N-(4-Bromophenyl)-2-chloroacetamide 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinolineRelated products of tetrahydrofuran

You may like
-
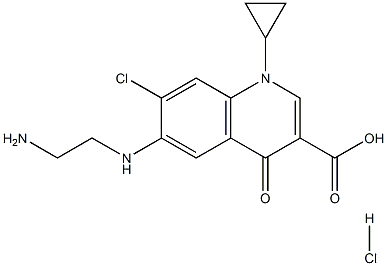
 226903-07-7 Ciprofloxacin EP Impurity F 99%View Details
226903-07-7 Ciprofloxacin EP Impurity F 99%View Details
226903-07-7 -
 CIPROFLOXACIN EP IMPURITY F 226903-07-7 90 % AboveView Details
CIPROFLOXACIN EP IMPURITY F 226903-07-7 90 % AboveView Details
226903-07-7 -
 Ciprofloxacin Impurity-F 226903-07-7 NLT 95%View Details
Ciprofloxacin Impurity-F 226903-07-7 NLT 95%View Details
226903-07-7 -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
Statement: All products displayed on this website are only used for non medical purposes such as industrial applications or scientific research, and cannot be used for clinical diagnosis or treatment of humans or animals. They are not medicinal or edible.
