4-Nitrophthalonitrile
- CAS NO.:31643-49-9
- Empirical Formula: C8H3N3O2
- Molecular Weight: 173.13
- MDL number: MFCD00040301
- EINECS: 250-748-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-05-12 09:22:35

What is 4-Nitrophthalonitrile?
Chemical properties
light yellow, light greenish or light grey to
The Uses of 4-Nitrophthalonitrile
4-Nitrophthalonitrile is a useful chemical in organic synthesis. Dyes and metabolites.
What are the applications of Application
4-Nitrophthalonitrile is used in organic synthesis to prepare compounds such as 4-Hydroxyphthalic anhydride, 4-hydroxyphthalonitrile, 4,4′-oxybis-(phthalonitrile), tetracyano diphenyl ether and phenol.
Preparation
Synthesis of 4-Nitrophthalonitrile: SOCl2 (83.5 mL. 1.144 mol) was added dropwise under nitrogen purge to dry DMF (200 mL) which had been cooled to 0-5 °C. The solution was allowed to stir for 15 min at 0-5 °C. The 4-nitrophthalamide (60.1 g, 0.286 mol) was then added and the solution was allowed to slowly warm to room temperature and react for 18 h under nitrogen purge. The solution was then slowly added to ice water to crystallize and precipitate the product. The 4-nitrophthalonitrile was collected using vacuum filtration, washed with ice cold water, and allowed to air dry overnight; yield: 45.2 g (92%); m.p.: 141 °C (det. by DSC)
1 H NMR((CD3)2SO): 8.41 (dd, 1H), 8.67 (dd, 1H), 9.03 (dd, 1H) FTIR: 3091 (m, aromatic C-H stretch), 2242 (d, CN stretch), 1534 (s, asymmetric N=O stretch), 1349 (s, symmetric N=O stretch), 853 (s, C-N stretch)
Synthesis
4-Nitrophthalonitrile synthesized from phthalimide in three steps. The reaction time of ruthenium chloride and HZSM-5 catalysts was very shorter than ammonium molybdate and Hβ catalysts. The yield while we used ruthenium chloride and HZSM-5 catalysts were very higher than another.
In a three necked flask, 70 mL of dry dimethylformamide (DMF) was cooled to 0 °C under a stream of nitrogen and 7.3 mL of thionyl chloride was added so that the internal temperature did not go beyond 5 °C. After addition, nitrogen flow was ceased and a calcium chloride tube was added to the top of flask. Meanwhile, the color of the medium was observed to be yellow. Then, 10 g (0.048 mol) of 4- nitrophthalamide was slowly added so that the internal temperature did not go beyond 5 °C. The mixture was stirred over ice bath for 1 hour. The mixture was stirred at room temperature for 2 hours and then poured over 500 g of ice-water. The precipitate was filtered and washed successively with water, 250 mL 5% sodium hydrogencarbonate solution, and water again and dried in a vacuum oven at 110-120 °C. Molecular formula: C8H3N3O2. Yield: 7.4 g (90%). Mp: 141 °C.
Properties of 4-Nitrophthalonitrile
| Melting point: | 142-144 °C(lit.) |
| Boiling point: | 303.75°C (rough estimate) |
| Density | 1.4553 (rough estimate) |
| refractive index | 1.6500 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| form | Crystalline Powder, Crystals and/or Chunks |
| color | Light yellow, light greenish or light gray to beige |
| Water Solubility | Sparingly soluble in water.(0.26 g/L) (25°C), |
| BRN | 1877554 |
| CAS DataBase Reference | 31643-49-9(CAS DataBase Reference) |
| EPA Substance Registry System | 1,2-Benzenedicarbonitrile, 4-nitro- (31643-49-9) |
Safety information for 4-Nitrophthalonitrile
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 4-Nitrophthalonitrile
4-Nitrophthalonitrile manufacturer
ASM Organics
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 5-Bromo-2-Fluoropyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 4-Bromo-3-(ethoxymethyl)benzoic acid 2,4-Dichloro-5-methoxyaniline 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine N2-Isobutyryl-2'-O-methylguanosine 2-chloro-5-methylpyridin-4-amine 4-Bromobenzaldehyde 2-Chloro-6-nitro benzothiazole 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amineRelated products of tetrahydrofuran
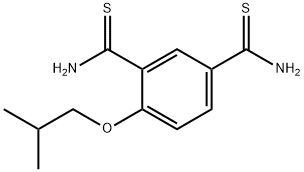
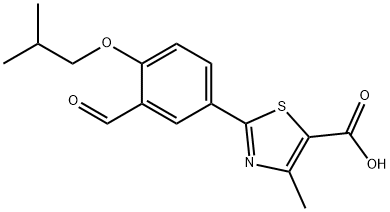
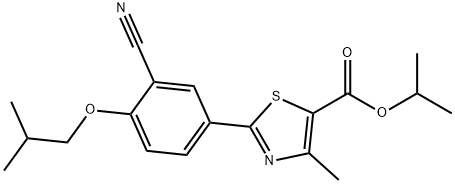
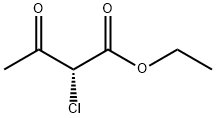
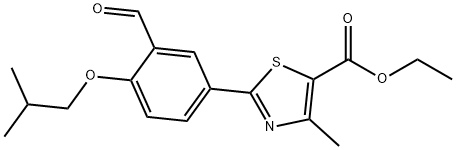
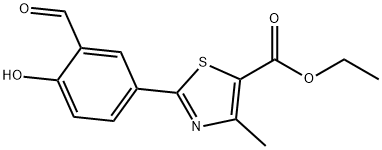
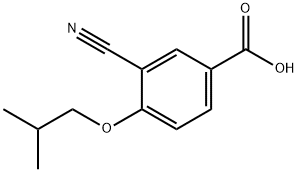
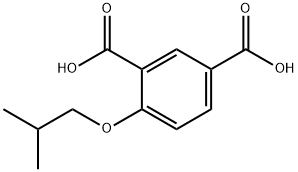
You may like
-
 31643-49-9 98%View Details
31643-49-9 98%View Details
31643-49-9 -
 4-Nitrophthalonitrile 98%View Details
4-Nitrophthalonitrile 98%View Details
31643-49-9 -
 4-Nitrophthalonitrile CAS 31643-49-9View Details
4-Nitrophthalonitrile CAS 31643-49-9View Details
31643-49-9 -
 4-Nitrophthalonitrile, 99% CAS 31643-49-9View Details
4-Nitrophthalonitrile, 99% CAS 31643-49-9View Details
31643-49-9 -
 4-Nitrophthalonitrile CAS 31643-49-9View Details
4-Nitrophthalonitrile CAS 31643-49-9View Details
31643-49-9 -
 4 Nitro PhthalonitrileView Details
4 Nitro PhthalonitrileView Details
31643-49-9 -
 4-NITROPHTHALONITRILE apiView Details
4-NITROPHTHALONITRILE apiView Details
31643-49-9 -
 4-Nitro PhthalonitrileView Details
4-Nitro PhthalonitrileView Details
31643-49-9
