2,3-Diaminobenzoic acid
- CAS NO.:603-81-6
- Empirical Formula: C7H8N2O2
- Molecular Weight: 152.15
- MDL number: MFCD00137818
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-04-24 14:58:19
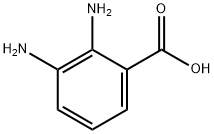
What is 2,3-Diaminobenzoic acid?
The Uses of 2,3-Diaminobenzoic acid
2,3-Diaminobenzoic acid (23DBA) is an important organic reagent mainly used in scientific research and laboratory experiments. 23DBA can undergo polycondensation with terephthalic acid dichloro anhydride[1]. 23DBA isomer is also used in the study of luminescence mechanism of red luminescent carbon dots (RCDs)[2]. In addition, 2,3-Diaminobenzoic acid also possesses antioxidant properties that help reduce the formation of free radicals in the body. It is also used as a chelating agent that effectively binds to metal ions and prevents them from forming insoluble complexes.
What are the applications of Application
2,3-Diaminobenzoic acid is an aromatic building block
Synthesis
2,3-Diaminobenzoic acid is synthesised using 3-nitroanthranilic acid as a raw material by chemical reaction. The specific synthesis steps are as follows:
2-amino-3-nitrobenzoic acid (9.36 g, 51.4 mmol) was dissolved in MeOH (100 mL) and there was added 10% Pd/C (2.0g). The reaction was stirred under H2-atmosphere (ambient pressure) until absorption ceased. The reaction was filtered through a pad of Celite and the filtrate was evaporated under reduced pressure. The product was purified by flash chromatography (CH2Cl2/MeOH 5:1) and the collected fractions were evaporated to give 2,3-Diaminobenzoic acid (5.40 g, 69%) as a dark brown solid. mp. 199 °C dec. (Litt4. 201 °C dec.). 1H NMR (300 MHz, DMSO-d6) δ 7.09 (dd, J = 8.0, 1.5 Hz, 1H), 6.67 (dd, J = 7.4, 1.5 Hz, 1H), 6.34 (dd, J = 8.0, 7.4 Hz, 2H), 6.32 (br s, 4H). 13C NMR (75 MHz, DMSO-d6) δ 170.5, 139.6, 135.6, 119.4, 117.0, 114.9, 110.3.
References
[1] V. N. ODNORALOVA. The polycondensation reaction of 2,4-diaminobenzoic acid with dichloroanhydride of terephthalic acid[J]. Fibre Chemistry, 1974. DOI:10.1007/BF00542946.
[2] LIU Y, DING H, ZHANG S, et al. Photoluminescence mechanism of red emissive carbon dots from a diaminobenzoic acid isomer?[J]. Materials Advances, 2023. DOI:10.1039/D3MA00836C.
Properties of 2,3-Diaminobenzoic acid
| Melting point: | 198-204 °C |
| Boiling point: | 274.61°C (rough estimate) |
| Density | 1.2804 (rough estimate) |
| refractive index | 1.5200 (estimate) |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| pka | 5.10±0.10(Predicted) |
| form | solid |
| color | Brown to dark brown |
| CAS DataBase Reference | 603-81-6(CAS DataBase Reference) |
Safety information for 2,3-Diaminobenzoic acid
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for 2,3-Diaminobenzoic acid
New Products
Pentadecanoic acid 3-Bromophenylacetic acid 3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Hendecanoic acid 2-Amino-5-cyanopyridine 2-Bromo-5-cyanopyridine 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 3′-Hydroxyacetophenone 5-fluoro-1,3-benzodioxole 5-Bromo-4-chloro-3-indolyl-β-D-galactopyranoside Tetracaine Hydrochloride Ep Grade 2,2,2-Trichloroethyl chloroformate 2-Bromo-6-fluoroaniline 2-Amino-4-phenyl-thiazole 4-Chloro-2-methyl quinoline 2-(Chloromethyl) quinazolin-4(3H)-one 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole N-(4-Bromophenyl)-2-chloroacetamide 6-Isopropylpicolinonitrile N-Methyl-3,5-dinitro-2-pyridinamine Phenyl-boronic acid-d5 10,11-Dihdyro-10-oxo-5H-dibenz[b,f]azepine-5-carboxamide-d4 5-(2,4-difluorophenyl)oxazolidin-2-oneRelated products of tetrahydrofuran
You may like
-
 2,3-Diamino benzoic acid, 274 Degree C, Packaging Type: BagView Details
2,3-Diamino benzoic acid, 274 Degree C, Packaging Type: BagView Details
603-81-6 -
 Deuterated Styrene-d8 97-99%View Details
Deuterated Styrene-d8 97-99%View Details
19361-62-7 -
![1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 3-bromo-[1,1'-biphenyl]-2-amine 97-99%View Details
1620885-59-7 -
 1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 6-Chloro-3,4-dimethylpyridin-2-amine 97-99%View Details
1503408-77-2 -
![665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 ethyl 1-(2,4-dichlorophenyl)-4,5-dihydro-1H-benzo[g]indazole-3-carboxylate 97-99%View Details
665020-24-6 -
 1187989-89-4 97-99%View Details
1187989-89-4 97-99%View Details
1187989-89-4 -
 Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
Methyl 1-(2,2,2-trifluoroethyl)azetidine-3-carboxylate 97-99%View Details
1872090-65-7 -
 32659-49-7 97-99%View Details
32659-49-7 97-99%View Details
32659-49-7
