Supplier Type
- Manufacturer
- Trader
- contract manufacturer
- Reagent
- custom synthesis
Supplier Region
- Gujarat(36)
- Bangalore(3)
- Hyderabad(25)
- Maharashtra(32)
- Delhi(13)
- Karnataka(7)
- Ahmedabad(19)
- Vadodara(10)
- New Delhi(9)
- Pune(4)
- Mumbai(47)
- Andhra Pradesh(2)
- Beijing(3)
- Kolkata(4)
- Uttar Pradesh(4)
- Haryana(2)
- Punjab(3)
- Telangana(2)
- Tamil Nadu(3)
- Rajasthan(1)
- Nagpur(1)
- Kerela(1)
Purity
- Salicylic Acid
- Rasagiline Mesylate Impurity C
- NLT 95%
- NLT 90%
- more than 95%
- more than 90%
- Mesalazine EP Impurity H
- Mathadone (M225865) impurity. Used in synthesis of methadone, antispasmodics and other pharmaceutica
- Magnesium content Min. 14%
- Heat resistant injection stage
- Greater than 99%
- Dipropargyl r- amino indane
- Dapagliflozin Intermediate, Dapagliflozin Impurity
- API Impurity / In-house working standard
- Acetylsalicylic Acid EP Impurity C
- ABOVE 99%
- 99.98%
- 99.95%
- 99.91%
- 99.90%
- 99.86%
- 99.85%
- 99.80%
- 99.73%
- 99.55%
- 99.50%
- 99.5% min
- 99.5%
- 99.42%
- 99.29%
- 99.26%
- 99.17%
- 99% Min
- > 99%
- 99%
- >99%
- 99 %
- 98-99%
- 98+
- 98.70%
- 98.60%
- 98.58%
- 98.57%
- 98.42%
- 98.33%
- 98.31%
- 98.12%
- 98.00%
- 98% Min
- >98%
- 98%
- 98 %
- 97-99%
- 97.57%
- 97.00%
- 97%
- >97%
- 97 %
- 96.80%
- 96.40%
- 96.39%
- 96.00%
- 96%
- 95.00%
- 95%
- >95%
- 94.00%
- >92%
- 91.21%
- >90%
- 90%
- 90 % Above
- 25 MG 50 MG 100 MG 250 MG EXTRA
- 100 +/- 5%
- (R)-N,N-di(prop-2-yn-1-yl)-2,3- dihydro-1
Package
- 0MG TO GRAM
- 25mg
- 50mg
- 100mg
- 250mg
- 500mg
- 1gm
- 1g
- 5g
- 10g
- 25G
- 50g
- 100g
- 500g
- 1000g
- 1kg
- 5kg
- 10kg
- 100kg
- 500kg
- 1and gm
- 95NLT %
- 2.5501e+00925 MG 50 MG 100 MG 250 MG EXTRA
- 25kg
- 50kg
- 1MT
- 250250 mg.
-
Sitagliptin Impurity 27
1608491-98-0
-
BLDAVCZELRQXFP-OWOJBTEDSA-N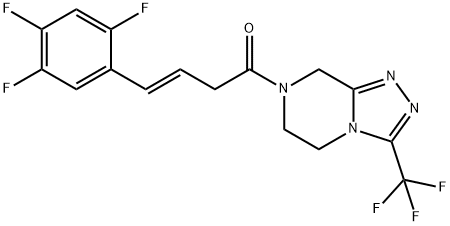
1803026-58-5
-
Sitagliptin DeaMino IMpurity 1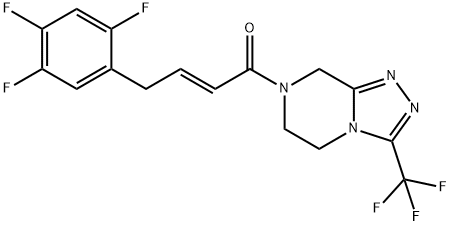
1253056-18-6
-
Sitagliptin Impurity 21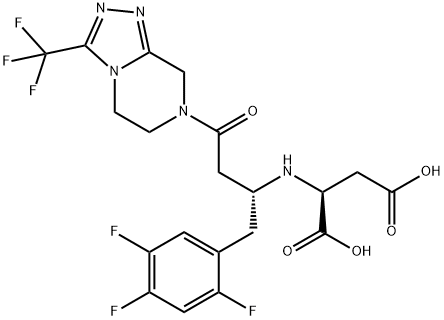
2088771-60-0
-
(2Z)-4-Oxo-4-[3-(trifluoromethyl)-5,6-dihydro-[1,2,4]triazolo[4,3-a]pyrazine-7(8H)-yl]-1-(2,4,5-trifluorophenyl)butan-2-one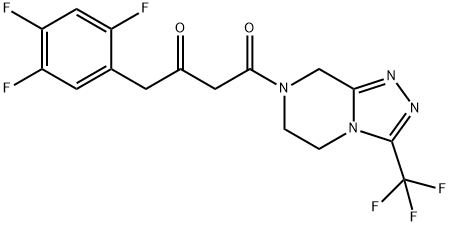
764667-65-4
-
7-Azabicyclo[4.1.0]hept-3-ene-3-carboxylic acid, 7-(1,1-dimethylethyl)-5-(1-ethylpropoxy)-, ethyl ester, (1R,5R,6R)-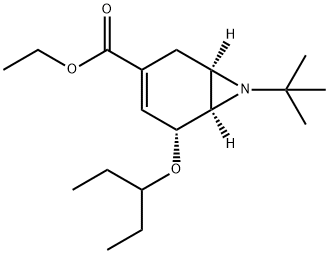
2417645-75-9
-
[1,1'-Biphenyl]-2-carboxamide, 4'-(azidomethyl)-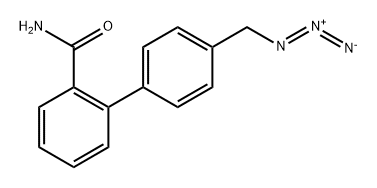
2763750-28-1
-
4-HYDROXY-N,N,2-TRIMETHYL-1H-BENZO[D]IMIDAZOLE-6-CARBOXAMIDE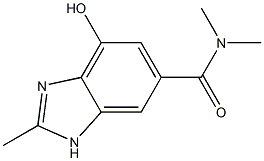
2168520-25-8
-
Carfilzomib Impurity 14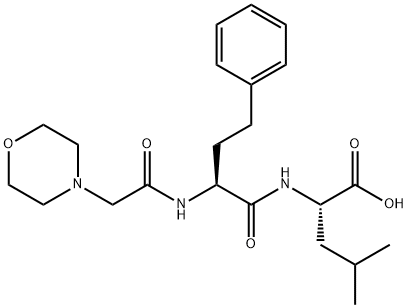
1396006-47-5
-
1,2-Cyclopentanediol, 3-[7-[[(1R,2S)-2-(3,4-difluorophenyl)cyclopropyl]amino]-5-[(R)-propylsulfinyl]-3H-1,2,3-triazolo[4,5-d]pyrimidin-3-yl]-5-(2-hydroxyethoxy)-, (1S,2S,3R,5S)-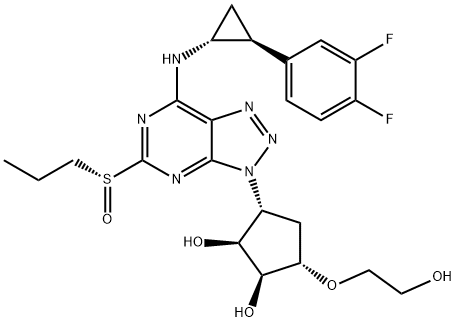
2417798-14-0
SITAGLIPTIN IMPURITY F 1608491-98-0
Sitagliptin FP Impurity C 1803026-58-5
Carfilzomib Impurity 14 (SS) 1396006-47-5

Nebivolol 118457-14-0

Loxoprofen 68767-14-6

Maltyl isobutyrate 65416-14-0

Sitagliptin 486460-32-6

3-Chloroperoxybenzoic acid 937-14-4

Sodium chloride 7647-14-5




