Supplier Type
- Manufacturer
- Reagent
- Trader
Supplier Region
- Gujarat(5)
- Maharashtra(3)
- Bangalore(1)
- Hyderabad(7)
- Pune(2)
- Mumbai(9)
- Ahmedabad(3)
- Karnataka(1)
- Punjab(2)
- Nagpur(1)
Purity
- Salicylic Acid
- Rasagiline Mesylate Impurity C
- NLT 95%
- NLT 90%
- more than 95%
- more than 90%
- Mesalazine EP Impurity H
- Mathadone (M225865) impurity. Used in synthesis of methadone, antispasmodics and other pharmaceutica
- Greater than 99%
- Empagliflozin intermediate, Dapagliflozin Impurity
- Dipropargyl r- amino indane
- Dapagliflozin Intermediate, Dapagliflozin Impurity
- API Impurity / In-house working standard
- Acetylsalicylic Acid EP Impurity C
- 99.99%
- 99.98%
- 99.95%
- 99.91%
- 99.86%
- 99.85%
- 99.80%
- 99.73%
- 99.42%
- 99.29%
- 99%
- 98.60%
- 98.12%
- 98%
- 97-99%
- 97.57%
- 97%
- 97 %
- 96.80%
- 96.39%
- 96.14%
- 96%
- 95.48%
- 95.00%
- 95%
- >95%
- 94.00%
- >92%
- 90%
- >90%
- 90 % Above
- 25 MG 50 MG 100 MG 250 MG EXTRA
- (R)-N,N-di(prop-2-yn-1-yl)-2,3- dihydro-1
Package
- 100mg
- 1g
- 25g
- 1kg
- 5kg
- 10kg
- 25kg
- 1MT
- 2.5501e+00925 MG 50 MG 100 MG 250 MG EXTRA
- 25mg
- 50mg
- 250mg
- 500mg
- 1gm
- 5g
- 1and gm
- 95NLT %
- 250250 mg.
-
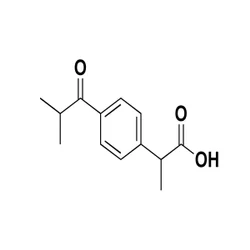
OPEN RING AZTREONAM
87500-74-1
-
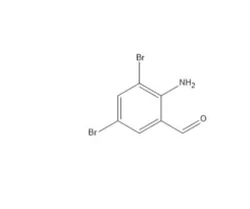
Aztreonam Impurity B
102586-36-7
-
OPEN RING DESULFATED AZTREONAM (E-ISOMER) / AZTREONAM USP RELATED COMPOUND B
-
Daptomycin Lactone Open Ring Impurity
-
Rivaroxaban Impurity H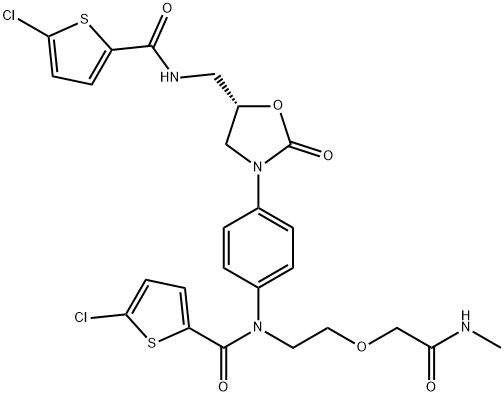
1807455-76-0
-
Everolimus Related Compound 2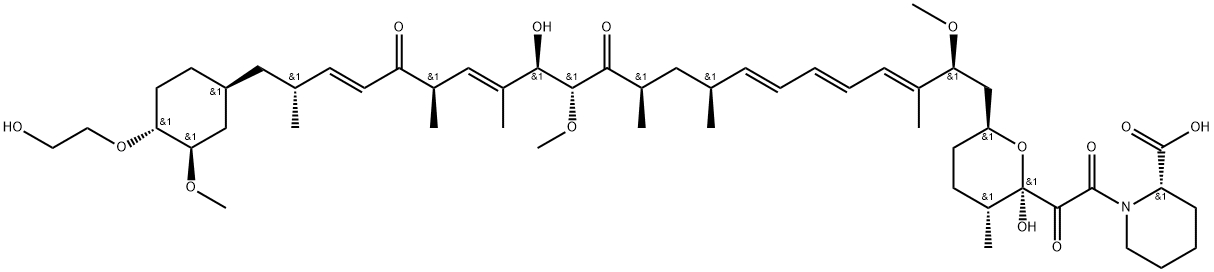
1062122-63-7
-
Rivaroxaban iMpurity A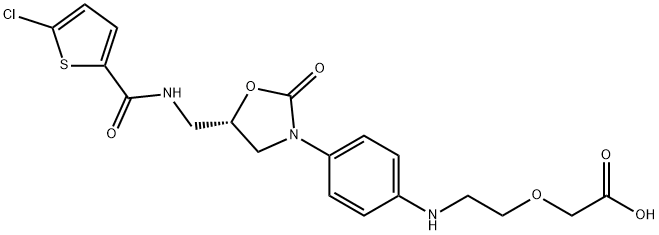
931204-39-6
-
Minocycline hydrochloride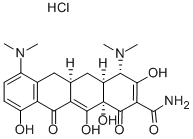
13614-98-7
-
Etoposide Impurity 9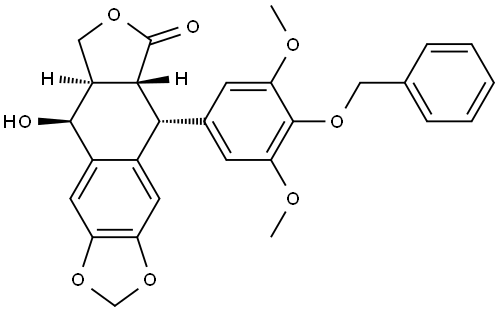
403642-80-8
-
Darunavir Amine dimer impurity
OPEN RING AZTREONAM 87500-74-1
Daptomycin Lactone Open Ring Impurity
Rivaroxaban Open-Ring N-Methyl Impurity 1807455-76-0
Everolimus -19-ere - open ring/Everolimus - Impurity B 1062122-63-7
Irinotecan Impurity L (HCl Salt) 1992961-26-8
Milestone Transparent Satellite Ring Container Big, For Kitchen Storage, Capacity: 1-3 Ltr