946524-26-1
COMPUTED DESCRIPTORS
| Molecular Weight | 302.36 g/mol |
|---|---|
| XLogP3 | 1.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 8 |
| Exact Mass | 302.17293854 g/mol |
| Monoisotopic Mass | 302.17293854 g/mol |
| Topological Polar Surface Area | 96.2 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 343 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
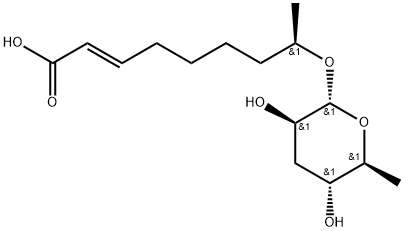
Ascr#3 is an (omega-1)-hydroxy fatty acid ascaroside obtained by formal condensation of the alcoholic hydroxy group of (2E,8R)-8-hydroxynon-2-enoic acid with ascarylopyranose (the alpha anomer). It is a major component of the dauer pheromone, used by the nematode Caenorhabditis elegans as a population-density signal to promote entry into an alternate larval stage, the nonfeeding and highly persistent dauer diapause, and also synergises with ascr#2, ascr#4, and ascr#8 in male attraction. It has a role as a Caenorhabditis elegans metabolite and a pheromone. It is an alpha,beta-unsaturated monocarboxylic acid and an (omega-1)-hydroxy fatty acid ascaroside. It is functionally related to a (2E,8R)-8-hydroxynon-2-enoic acid. It is a conjugate acid of an ascr#3(1-).
