COMPUTED DESCRIPTORS
| Molecular Weight | 755.0 g/mol |
|---|---|
| XLogP3 | 3.5 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 9 |
| Exact Mass | 754.48672766 g/mol |
| Monoisotopic Mass | 754.48672766 g/mol |
| Topological Polar Surface Area | 199 Ų |
| Heavy Atom Count | 53 |
| Formal Charge | 0 |
| Complexity | 1320 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 19 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
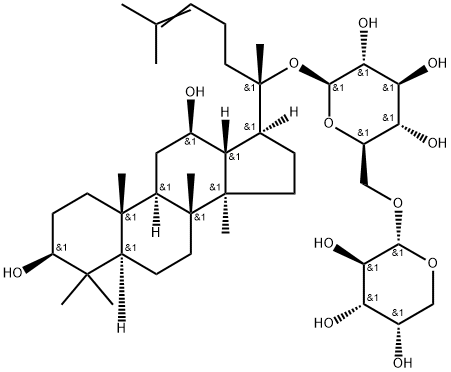
Ginsenoside C-Y is a ginsenoside found in Panax species that is dammarane which is substituted by hydroxy groups at the 3beta, 12beta and 20 pro-S positions, in which the hydroxy group at position 20 has been converted to the corresponding alpha-L-arabinopyranosyl-(1->6)-beta-D-glucopyranoside, and in which a double bond has been introduced at the 24-25 position. It has a role as a plant metabolite. It is a 12beta-hydroxy steroid, a 3beta-hydroxy steroid, a beta-D-glucoside, a disaccharide derivative, a ginsenoside, a tetracyclic triterpenoid and a 3beta-hydroxy-4,4-dimethylsteroid. It derives from a hydride of a dammarane.