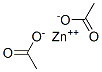CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Obtained in both anhydrous form and as a dihydrate. Both are white crystalline solids. The primary hazard is the threat posed to the environment. Immediate steps should be taken to limit spread to the environment. Used to preserve wood, to make other zinc compounds, as a food and feed additive. |
|---|---|
| Color/Form | Exists in two crystalline forms, alpha (wurtzite) and beta (sphalerite) |
| Solubility | Solubility: 40 g/ 100 g water at 25 °C; 67 g/100 g water at 100 °C; 3 g/ 100 g alcohol at 25 °C /Zinc acetate dihydrate/ |
| Density | 1.74 at 68 °F (USCG, 1999) - Denser than water; will sink |
| Decomposition | When heated to decomposition it emits toxic fumes of /zinc oxide/. |
| pH | Between 6,0 and 8,0 (5 % solution) |
| Other Experimental Properties | Changes to alpha form at 1020 °C; sublimes at 1180 °C /beta/ |
| Chemical Classes | Metals -> Organic Acids, Metal Salts |
COMPUTED DESCRIPTORS
| Molecular Weight | 183.5 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 181.955750 g/mol |
| Monoisotopic Mass | 181.955750 g/mol |
| Topological Polar Surface Area | 80.3 Ų |
| Heavy Atom Count | 9 |
| Formal Charge | 0 |
| Complexity | 25.5 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 3 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Obtained in both anhydrous form and as a dihydrate. Both are white crystalline solids. The primary hazard is the threat posed to the environment. Immediate steps should be taken to limit spread to the environment. Used to preserve wood, to make other zinc compounds, as a food and feed additive.