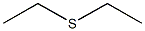CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Diethyl sulfide appears as a colorless oily liquid with a garlic-like odor. Less dense than water. Vapors heavier than air. May irritate skin and eyes. Used to make other chemicals. |
|---|---|
| Color/Form | Liquid |
| Odor | Garlic-like odor |
| Boiling Point | 92.1 °C |
| Melting Point | -103.9 °C |
| Flash Point | -6.7 °C |
| Solubility | Soluble in ethanol, diethyl ether; slightly soluble in carbon tetrachloride. |
| Density | 0.8362 g/cu cm at 20 °C |
| Vapor Density | 3.11 (AIR= 1) |
| Vapor Pressure | 60.2 [mmHg] |
| LogP | log Kow = 1.95 |
| Henry's Law Constant | Henry's Law constant = 1.68X10-3 atm-cu m/mol at 25 °C |
| Decomposition | Hazardous decomposition products formed under fire conditions: Carbon oxides, Sulfur oxides |
| Odor Threshold | Odor Threshold Low: 0.0008 [mmHg] Odor Threshold High: 0.0054 [mmHg] [HSDB] Odor threshold from CHEMINFO |
| Refractive Index | Index of refraction: 1.4430 at 20 °C/D |
| Kovats Retention Index | 698 692 692 693 693 690 704 698 693 690 683 685 |
| Other Experimental Properties | Conversion factor: 3.68 mg/cu m = 1 ppm |
| Chemical Classes | Other Classes -> Sulfur Compounds |
COMPUTED DESCRIPTORS
| Molecular Weight | 90.19 g/mol |
|---|---|
| XLogP3 | 1.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Exact Mass | 90.05032149 g/mol |
| Monoisotopic Mass | 90.05032149 g/mol |
| Topological Polar Surface Area | 25.3 Ų |
| Heavy Atom Count | 5 |
| Formal Charge | 0 |
| Complexity | 11.1 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Diethyl sulfide appears as a colorless oily liquid with a garlic-like odor. Less dense than water. Vapors heavier than air. May irritate skin and eyes. Used to make other chemicals.