CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Tannic acid is a light yellow to tan solid with a faint odor. Sinks and mixes with water. (USCG, 1999) |
|---|---|
| Color/Form | YELLOWISH-WHITE TO LIGHT BROWN, AMORPHOUS, BULKY POWDER OR FLAKES, OR SPONGY MASSES |
| Odor | FAINT CHARACTERISTIC ODOR |
| Taste | ASTRINGENT TASTE |
| Melting Point | 200 °C |
| Flash Point | 390 °F |
| Solubility | VERY SOL IN ALCOHOL, ACETONE; INSOL IN ETHER, BENZENE, CHLOROFORM & CARBON DISULFIDE |
| Density | greater than 1 at 68 °F (USCG, 1999) |
| Stability/Shelf Life | AQ SOLN SHOULD BE FRESHLY PREPARED BECAUSE THEY ARE UNSTABLE DUE TO BACTERIAL ACTION & IN PRESENCE OF LIGHT & OXYGEN. |
| Autoignition Temperature | 980 °F (USCG, 1999) |
| Decomposition | When heated to decomp ... emits acrid smoke and fumes. |
| Other Experimental Properties | AQ SOLN DECOMP ON HEATING; REPORTED TO CATALYSE FORMATION OF NITROSODIETHYLAMINE |
| Chemical Classes | UVCB |
COMPUTED DESCRIPTORS
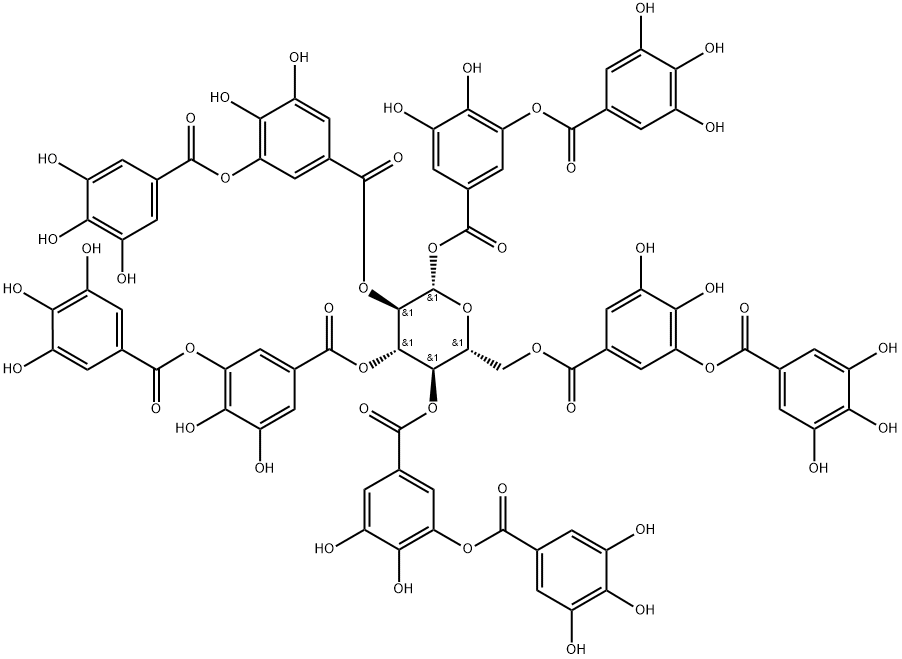
| Molecular Weight | 1701.2 g/mol |
|---|---|
| XLogP3 | 6.2 |
| Hydrogen Bond Donor Count | 25 |
| Hydrogen Bond Acceptor Count | 46 |
| Rotatable Bond Count | 31 |
| Exact Mass | 1700.1729741 g/mol |
| Monoisotopic Mass | 1700.1729741 g/mol |
| Topological Polar Surface Area | 778 Ų |
| Heavy Atom Count | 122 |
| Formal Charge | 0 |
| Complexity | 3570 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Tannic acid is a light yellow to tan solid with a faint odor. Sinks and mixes with water. (USCG, 1999)